
��ͼ��ijθҩ��ǩ�ϵIJ������ݣ������ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ10Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ25.8g������㣺
��1��10ƬҩƷ������Ϊ g����Ӧ���������������Ϊ g��
��2��ҩƷ��̼�����Ƶ�����������
��3����Ӧ��������Һ�����ʵ�������������������������0.1%��

1��5 2.2
��2����3���⣺��Ҫ����2.2g������̼��Ҫ�μӷ�Ӧ��̼����������Ϊx��
�����Ȼ�������Ϊy����
NaHCO3+HCl ═ NaCl + H2O+ CO2��
84 58.5 44
x y 2.2g
 ���x=4.2t y=2.925g
���x=4.2t y=2.925g
ҩƷ��̼�����Ƶ�����������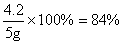
��Ӧ��������Һ����Ϊ��4.2g+23g-2.2g=25g����Ӧ��������Һ�����ʵ�����������
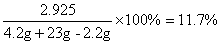
�𣺣�2��ҩƷ��̼�����Ƶ���������84%��
��3����Ӧ��������Һ�����ʵ���������11.7%



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��2012?��������ͼ��ijθҩ��ǩ�ϵIJ������ݣ������ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ10Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ25.8g������㣺
��2012?��������ͼ��ijθҩ��ǩ�ϵIJ������ݣ������ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ10Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ25.8g������㣺�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��ͼ��ijθҩ��ǩ�ϵIJ������ݣ�ij��ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ20Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ28.6g��
��ͼ��ijθҩ��ǩ�ϵIJ������ݣ�ij��ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ20Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ28.6g���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ�п����� ���ͣ�������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ͼ��ijθҩ��ǩ�ϵIJ������ݣ������ѧ��ѧ��ȤС�����ⶨ��ҩƷ��̼�����ƣ�NaHCO3��������������ȡ10Ƭ��ҩƷ��������ձ��У������ձ��еμ�ϡ������ǡ����ȫ��Ӧ�����ʲ�����ˮ��Ҳ���μӷ�Ӧ����������ϡ����23g����÷�Ӧ���ձ������ʵ�������Ϊ25.8g������㣺
��1��10ƬҩƷ������Ϊ g����Ӧ���������������Ϊ g��
��2��ҩƷ��̼�����Ƶ�����������
��3����Ӧ��������Һ�����ʵ�������������������������0.1%��

�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com