
| ¼ÓČėĻ”ŃĪĖįµÄ“ĪŹż | µŚŅ»“Ī | µŚ¶ž“Ī | µŚČż“Ī | µŚĖÄ“Ī | µŚĪå“Ī |
| ÉÕ±¼°·“Ó¦ŗó»ģŗĻĪļµÄ×ÜÖŹĮæ/g | 122.2 | 146.1 | 170.0 | 193.9 | 218.9 |
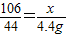 £¬
£¬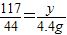
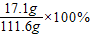 ”Ö15.3%£®
”Ö15.3%£®

 æĪŹ±ŃµĮ·½ĖÕČĖĆń³ö°ęÉēĻµĮŠ“š°ø
æĪŹ±ŃµĮ·½ĖÕČĖĆń³ö°ęÉēĻµĮŠ“š°ø »ĘøŌ¾µäȤĪ¶æĪĢĆĻµĮŠ“š°ø
»ĘøŌ¾µäȤĪ¶æĪĢĆĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ


µŲµć ĻīÄæ |
A | B | C |
| pH | 6 | 4 | 5 |
| 1LĖ®ÖŠ ČܽāµÄŃõĘų |
0.011g | 0.007g | 0.003g |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ0110 ŌĀæ¼Ģā ĢāŠĶ£ŗĢīæÕĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ2007-2008ѧğ½Ī÷Ź”¾°µĀÕņĄÖĘ½ŹŠŗ£øŪ֊ѧ¾ÅÄź¼¶£ØÉĻ£©ŌĀæ¼»ÆѧŹŌ¾ķ£ØµŚ1-8µ„ŌŖ£©£Ø½āĪö°ę£© ĢāŠĶ£ŗ½ā“šĢā

²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com