�ҹ�Ŀǰʹ�õ�ȼ����Ҫ��ú��ʯ�ͣ��������úȼ��ʱ���ɵ�SO2����Ⱦ������
��1��ij�������糧ÿ��ȼ�պ���1.6%��ú100t����ú�е���ȫ��ת��ΪSO2����ó�ÿ�����SO2______t��Ϊ��ֹSO3��Ⱦ���ɽ�SO2ͨ��ʯ����������Ca��HSO3��2����÷�Ӧ�Ļ�ѧ����ʽΪ______��
��2�����ұ��涨��ҵ������SO2�������ó���0.15mg/m3����ҵ�ϲ���SO2�ĺ���ʱ�����Ը��ݷ�Ӧ��SO2+2H2O+I2=H2SO4+2HI����ȡ�ó�������Ʒ1000L����0.0254%�ĵ⣨12����Һ2g����ȫ��Ӧ���Լ���ó��ŷŵķ�����SO2�ĺ����Ƿ���Ϲ��ұ���
���𰸡�
���������ݷ�Ӧ��������P�������غ㶨�ɿ�����ȷ����д��ѧ����ʽ��������Ԫ�ص������Ͷ�����������Ԫ�ص��������������Լ���ó�������������������ݷ�Ӧ�Ļ�ѧ����ʽ��������ó��ŷŵķ�����SO
2�ĺ����Ƿ���Ϲ��ұ���
����⣺��1��ÿ��ʹ��ú���е��������Ϊ100t×1.6%�����ڶ�����������Ԫ�ص���������Ϊ
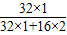
×100%=50%�����Ըó�ÿ������������������Ϊ��100t×1.6%÷50%=3.2t�����3.2��
����������������Ʒ�Ӧ��������������ƣ���Ӧ�Ļ�ѧ����ʽΪ��2SO
2+Ca��OH��
2�TCa��HSO
3��
2��2����������������ΪX��
SO
2+2H
2O+I
2=H
2SO
4+2HI
64 254
X 2g×0.0254%
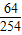
=
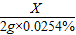
X=1.28×10
-4g
��l000L������SO
2������Ϊ1.28×10
-4g��
1.28×10
-4g=O.128mg/m
3��0.15mg/m
3�����Ϲ��ұ���
�����������Ҫ����������ط���ļ��㷽����ֻ����������ط���ļ��㷽������ͨ������ó���ȷ�Ĵ𰸣�
��Ŀ�����л�ѧ
��Դ��2003���ʮ���조��ԭ����ȫ������ѧ����ѧ���ʺ�ʵ���������������������������Ծ��������棩
���ͣ������
�ҹ�ʹ�õ�ȼ����Ҫ��ú̿��ʯ�ͣ��������ú̿ȼ��ʱ�ŷų���Ⱦ�������ж�������Ҫ��SO2����������ˮ��Ϊ���꽵�䵽���������ܴ�Σ������ش��������⣺
��1����ٳ����������Σ��ʵ����
�� ���� ���� ��
��2������ֲ���п�����Ⱦ�������ж�������������ڳ���ֲ���ж���Щ�����н�ǿ�������������� ����дֲ�����ƣ���
��3����SO2��Ⱦ����ʱ��Ŀǰ��һ����Ч�ķ���������ֱ��������̼��Ʒ�ĩ������Ч���ɴ�85%���仯ѧ����ʽΪ��2CaCO3+2SO2+O2�T2CaSO4+2CO2��ʵ��֤��ʯ�ҽ�[Ca��OH��2]�ڿ����п�����SO2����CaSO4���������ʣ���д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ
��Դ����7.3 ʹ��ȼ�϶Ի�����Ӱ�졷2010��ͬ����ϰ��1���������棩
���ͣ������
�ҹ�ʹ�õ�ȼ����Ҫ��ú̿��ʯ�ͣ��������ú̿ȼ��ʱ�ŷų���Ⱦ�������ж�������Ҫ��SO2����������ˮ��Ϊ���꽵�䵽���������ܴ�Σ������ش��������⣺
��1����ٳ����������Σ��ʵ����
�� ���� ���� ��
��2������ֲ���п�����Ⱦ�������ж�������������ڳ���ֲ���ж���Щ�����н�ǿ�������������� ����дֲ�����ƣ���
��3����SO2��Ⱦ����ʱ��Ŀǰ��һ����Ч�ķ���������ֱ��������̼��Ʒ�ĩ������Ч���ɴ�85%���仯ѧ����ʽΪ��2CaCO3+2SO2+O2�T2CaSO4+2CO2��ʵ��֤��ʯ�ҽ�[Ca��OH��2]�ڿ����п�����SO2����CaSO4���������ʣ���д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>

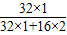 ×100%=50%�����Ըó�ÿ������������������Ϊ��100t×1.6%÷50%=3.2t�����3.2��
×100%=50%�����Ըó�ÿ������������������Ϊ��100t×1.6%÷50%=3.2t�����3.2��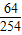 =
=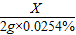


 �����Ƹ���ʦ����ϵ�д�
�����Ƹ���ʦ����ϵ�д� ��ͨ����ͬ����ϰ��ϵ�д�
��ͨ����ͬ����ϰ��ϵ�д� ����С����ͬ������ϵ�д�
����С����ͬ������ϵ�д�
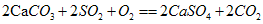 ��ʵ��֤��ʯ�ҽ�[Ca(OH)2]�ڿ�����Ҳ������SO2��������ƺ��������ʣ�д���÷�Ӧ�Ļ�ѧ����ʽ��________________________________��
��ʵ��֤��ʯ�ҽ�[Ca(OH)2]�ڿ�����Ҳ������SO2��������ƺ��������ʣ�д���÷�Ӧ�Ļ�ѧ����ʽ��________________________________��