

 ĆūÅĘ֊ѧæĪŹ±×÷ŅµĻµĮŠ“š°ø
ĆūÅĘ֊ѧæĪŹ±×÷ŅµĻµĮŠ“š°ø Ć÷Ģģ½ĢÓżæĪŹ±ĢŲѵĻµĮŠ“š°ø
Ć÷Ģģ½ĢÓżæĪŹ±ĢŲѵĻµĮŠ“š°ø Õć½ŠĀæĪ³ĢČżĪ¬Äæ±ź²āĘĄæĪŹ±ĢŲѵĻµĮŠ“š°ø
Õć½ŠĀæĪ³ĢČżĪ¬Äæ±ź²āĘĄæĪŹ±ĢŲѵĻµĮŠ“š°ø ÖÜÖÜĒå¼ģ²āĻµĮŠ“š°ø
ÖÜÖÜĒå¼ģ²āĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
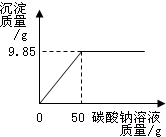
 Š”ŗģĶ¬Ń§ŌŚÄ³»Æ¹¤³§½ųŠŠÉē»įŹµ¼ł£¬¼¼ŹõŌ±ÓėŠ”ŗģŅ»Ęš·ÖĪöÓÉĀČ»Æ±µŗĶĀČ»ÆÄĘ×é³ÉµÄ²śĘ·ÖŠĀČ»ÆÄʵÄÖŹĮæ·ÖŹż£®Č”16.25g¹ĢĢåѳʷ£¬Č«²æČÜÓŚ143.6mLĖ®ÖŠ£¬ĻņĖłµĆµ½µÄ»ģŗĻČÜŅŗÖŠÖšµĪ¼ÓČėČÜÖŹÖŹĮæ·ÖŹżĪŖ10.6%µÄĢ¼ĖįÄĘČÜŅŗ£¬¼ĒĀ¼ĮĖČēĶ¼ĖłŹ¾µÄĒśĻß¹ŲĻµ£®
Š”ŗģĶ¬Ń§ŌŚÄ³»Æ¹¤³§½ųŠŠÉē»įŹµ¼ł£¬¼¼ŹõŌ±ÓėŠ”ŗģŅ»Ęš·ÖĪöÓÉĀČ»Æ±µŗĶĀČ»ÆÄĘ×é³ÉµÄ²śĘ·ÖŠĀČ»ÆÄʵÄÖŹĮæ·ÖŹż£®Č”16.25g¹ĢĢåѳʷ£¬Č«²æČÜÓŚ143.6mLĖ®ÖŠ£¬ĻņĖłµĆµ½µÄ»ģŗĻČÜŅŗÖŠÖšµĪ¼ÓČėČÜÖŹÖŹĮæ·ÖŹżĪŖ10.6%µÄĢ¼ĖįÄĘČÜŅŗ£¬¼ĒĀ¼ĮĖČēĶ¼ĖłŹ¾µÄĒśĻß¹ŲĻµ£®²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
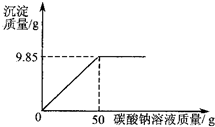 Š”ŗģĶ¬Ń§ŌŚÄ³»Æ¹¤³§½ųŠŠÉē»įŹµ¼ł£¬¼¼ŹõŌ±ÓėŠ”ŗģŅ»Ęš·ÖĪöÓÉĀČ»Æ±µŗĶĀČ»ÆÄĘ×é³ÉµÄ²śĘ·ÖŠĀČ»ÆÄʵÄÖŹĮæ·ÖŹż£®Č”16.25g¹ĢĢåѳʷ£¬Č«²æČÜÓŚ143.6mLĖ®ÖŠ£¬ĻņĖłµĆµ½µÄ»ģŗĻČÜŅŗÖŠÖšµĪ¼ÓČėČÜÖŹÖŹĮæ·ÖŹżĪŖ10.6%µÄĢ¼ĖįÄĘČÜŅŗ£¬¼ĒĀ¼ĮĖČēĶ¼ĖłŹ¾µÄĒśĻß¹ŲĻµ£®
Š”ŗģĶ¬Ń§ŌŚÄ³»Æ¹¤³§½ųŠŠÉē»įŹµ¼ł£¬¼¼ŹõŌ±ÓėŠ”ŗģŅ»Ęš·ÖĪöÓÉĀČ»Æ±µŗĶĀČ»ÆÄĘ×é³ÉµÄ²śĘ·ÖŠĀČ»ÆÄʵÄÖŹĮæ·ÖŹż£®Č”16.25g¹ĢĢåѳʷ£¬Č«²æČÜÓŚ143.6mLĖ®ÖŠ£¬ĻņĖłµĆµ½µÄ»ģŗĻČÜŅŗÖŠÖšµĪ¼ÓČėČÜÖŹÖŹĮæ·ÖŹżĪŖ10.6%µÄĢ¼ĖįÄĘČÜŅŗ£¬¼ĒĀ¼ĮĖČēĶ¼ĖłŹ¾µÄĒśĻß¹ŲĻµ£®²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ2008Äź½ĖÕŹ”ŠģÖŻĮłÖŠÖŠæ¼»ÆŃ§Ä£ÄāŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗ½ā“šĢā

²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com