
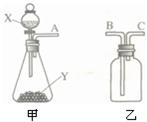
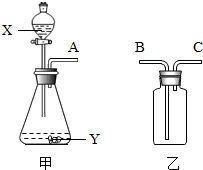
��ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��ֹ���װ�á�
 ��1�����Ʊ����������������Ҳ����������в���������
��1�����Ʊ����������������Ҳ����������в���������
�����壬���Լ�X����____ ___����ͼʾ���õ���װ���ռ�������AӦ��________���B����C����������
��2�����Ʊ���������������Y�Ǻ�ɫ��ĩ������з�Ӧ
�Ļ�ѧ����ʽΪ_______ _____��
��3����X��ϡ���ᣬY��ʯ��ʯ���üס���װ����ɷ��������顢�������ռ�CO2�����ϵ��װ�á���Ҫ���������̼���壬������Ӧʢװ ��������ʢװŨ���ᣬ��Ũ�����������____________________��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 33����ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��ֹ���װ�ã�
33����ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��ֹ���װ�ã��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
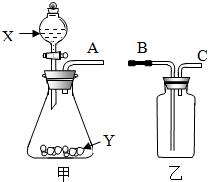 ��ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��ֹ���װ�ã�
��ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��ֹ���װ�ã��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
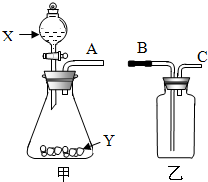 ��2012?�³�����ģ����ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��װ�ã�
��2012?�³�����ģ����ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��װ�ã��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
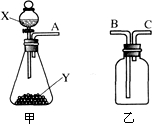 ��ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��ֹ���װ�ã���������и��⣮
��ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��ֹ���װ�ã���������и��⣮�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��2012?Т����ģ�⣩��ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��ֹ���װ�ã�
��2012?Т����ģ�⣩��ͼ��ʵ���ҳ��õ����������Ʊ����ռ��������Ķ��ֹ���װ�ã�
| ||
| ||
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com