
| ϡ��������� | ʣ���������� |
| ��һ�μ���10�� | 15�� |
| �ڶ��μ���10�� | 10�� |
| �������10�� | 5�� |
| ���Ĵμ���10�� | 2�� |
| 20g-2g |
| 20g |
| 100 |
| 44 |
| 9g |
| x |


 ����������ϵ�д�
����������ϵ�д� �Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
�Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ��2012?��������ģ��2011��9�£�ij���౻�غ����°���������������������ߵĽ��ǣ�����ͬѧ�ǿ�չ�˶�����ɷּ����õ�̽���������������ϵ�֪��
��2012?��������ģ��2011��9�£�ij���౻�غ����°���������������������ߵĽ��ǣ�����ͬѧ�ǿ�չ�˶�����ɷּ����õ�̽���������������ϵ�֪��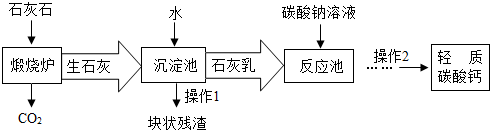
| ||
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| �Ϻ���Ч���� | �Ϻ��������� | ���Ӿ����� | |
| Ħ������ | �������� | ̼��� | �������� |
| Ħ������������� | �� �� |
�� �� |
������ ������ |
| 1���� |
| 2��ˮ |
| 3��̼�����Һ |
| ||
| ||
| ���� |
| ��ˮ |
| ͨ�������̼ |
| ���� |
| ��ˮ |
| ͨ�������̼ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�콭��ʡ�Ͼ������������꼶�п���ģ��ѧ�Ծ����������� ���ͣ�������
��(10��)2011��9�£�ij���౻�غ����°���������������������ߵĽ��ǣ�����ͬѧ�ǿ�չ�˶�����ɷּ����õ�̽���������������ϵ�֪��
��1���ܶ�Ʒ��������С����������������еġ�������ָ
A�������� B����Ԫ�� C����ԭ��
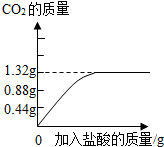
��2������ȥ����Ҫ��������Ħ�����á�ijƷ�������е�Ħ������̼��ƣ�Ϊ�˼��鲢�ⶨ������̼��Ƶ�����������ͬѧ��ȡ��10g���࣬��������ϡ���Ტ���衣ʵ������м�¼�����������γ���ͼ���ߣ�
��Ʒ��������̼��Ƶ���������Ϊ���١�
��3����������Ħ����������̼��ƿ���ʯ��ʯ���Ʊ�����ҵ����Ҫ�����������£�
�١�����¯���з�����Ӧ�Ļ�ѧ����ʽ�ǣ� ���ڡ���Ӧ�ء�����Ҫ������Ӧ�Ļ�ѧ����ʽ�ǣ� ��
�ڡ������ء�����ʯ����ˮ��ַ�Ӧ��ɵõ������dz�ϸС����ʯ�ҽ�����ʯ�ҽ�Ϊ
(ѡ�����Һ������Һ��������Һ��)��
�����˽�����������������CO2�����̼������Һ�������������ŵ��� ��
С���������Ϻ��֪��a��������̼����ͨ������������Һ�������·�Ӧ�� CO2+Ca(OH)2=CaCO3��+H2O�� CaCO3+ H2O + CO2=Ca(HCO3)2��
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
2011��9�£�ij���౻�غ����°���������������������ߵĽ��ǣ�����ͬѧ�ǿ�չ�˶�����ɷּ����õ�̽���������������ϵ�֪��
��1���ܶ�Ʒ��������С����������������еġ�������ָ
A�������� B����Ԫ�� C����ԭ��
��2������ȥ����Ҫ��������Ħ�����á�ijƷ�������е�Ħ������̼��ƣ�Ϊ�˼��鲢�ⶨ������̼��Ƶ�����������ͬѧ��ȡ��10g���࣬��������ϡ���Ტ���衣ʵ������м�¼�����������γ���ͼ���ߣ�
��Ʒ��������̼��Ƶ���������Ϊ���١�

|
|
|
 |
�١�����¯���з�����Ӧ�Ļ�ѧ����ʽ�ǣ� ���ڡ���Ӧ�ء�����Ҫ������Ӧ�Ļ�ѧ����ʽ�ǣ� ��
�ڡ������ء�����ʯ����ˮ��ַ�Ӧ��ɵõ������dz�ϸС����ʯ�ҽ�����ʯ�ҽ�Ϊ
(ѡ�����Һ������Һ��������Һ��)��
�����˽�����������������CO2�����̼������Һ�������������ŵ��� ��
С���������Ϻ��֪��a��������̼����ͨ������������Һ�������·�Ӧ��
CO2+Ca(OH)2=CaCO3��+H2O�� CaCO3+ H2O + CO2=Ca(HCO3)2��
|
Ca(HCO3)2===== CaCO3��+H2O+ CO2����
Ϊ�˷�ֹ���ø÷��Ƶõ�����̼����л���Ca(HCO3)2������2�б���Ҫ���е�һ�������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�꽭��ʡ�Ͼ����������п���ѧ��ģ�Ծ��������棩 ���ͣ������
 2011��9�£�ij���౻�غ����°���������������������ߵĽ��ǣ�����ͬѧ�ǿ�չ�˶�����ɷּ����õ�̽���������������ϵ�֪��
2011��9�£�ij���౻�غ����°���������������������ߵĽ��ǣ�����ͬѧ�ǿ�չ�˶�����ɷּ����õ�̽���������������ϵ�֪��
 CaCO3��+H2O+CO2����
CaCO3��+H2O+CO2�����鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com