
 愉快的寒假南京出版社系列答案
愉快的寒假南京出版社系列答案科目:初中化学 来源: 题型:
实验室制取二氧化碳一般分为五个步骤:①检查装置的气密性,②连接组装 仪器,③向长颈漏斗中注入稀盐酸, ④向锥形瓶中放入大理石, ⑤收集气体。正确的顺序是:
仪器,③向长颈漏斗中注入稀盐酸, ④向锥形瓶中放入大理石, ⑤收集气体。正确的顺序是:
A.①②③④⑤ B.①②④③⑤ C.②①③④⑤ D.②①④③⑤
查看答案和解析>>
科目:初中化学 来源: 题型:
雨水资源化是一种发展趋势。
(1)受酸性气体影响,雨水在降落过程中形成了酸雨。要测定某雨水酸碱度,可使用 。
(2)收集的地面雨水常携带树叶、泥沙等不溶性杂物,应进行沉淀、__________等处理;写出图中①号仪器的名称 ,②号仪器的作用是 。
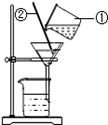
查看答案和解析>>
科目:初中化学 来源: 题型:
下列关于硝酸铵(NH4NO3)的说法,不正确的是
A.NH4NO3是一种常见的氮肥
B.NH4NO3中含2种不同的原子团
C.NH4NO3中各元素的质量比为1:4:1:3
 |
查看答案和解析>>
科目:初中化学 来源: 题型:
H7N9型禽流感是由H7N9亚型禽流感病毒引起的急性呼吸道传染病,可用ClO2进行环境消毒。制取ClO2的微观过程如图所示:

 | |||
 | |||
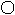 (其中: 表示钠原子, 表示氯原子, 表示氧原子)
(其中: 表示钠原子, 表示氯原子, 表示氧原子)
请回答下列问题:
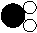
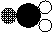 (1)如图所示的四种物质:
(1)如图所示的四种物质:
 |  |
a b c d
在a、b、c、d四种物质中,属于氧化物的是 (填字母)。
(2)制取ClO2的化学方程式为 。
查看答案和解析>>
科目:初中化学 来源: 题型:
某化学兴趣小组同学用Na2CO3溶液和浓HCl来研究简易灭火器的反应原理时,对废液的成分进行探究。
【推理假设】 废液的成分中一定有 ,可能有Na2CO3或盐酸.
【实验探究】 确定废液成分中是否含有Na2CO3或盐酸
请你协助兴趣小组的同学完成实验探究,并完成实验报告。
| 实验步骤 | 现象 | 结论 |
| 该废液中一定没有盐酸 | ||
| 该废液中一定含有Na2CO3 |
查看答案和解析>>
科目:初中化学 来源: 题型:
甲、乙、丙、丁都是含有碳元素的物质,它们之间有如下转化关系:
⑴甲+丙→ 乙 ⑵乙+O2→ 丙 ⑶丁在高温下分解可得到丙
则按甲、乙、丙、丁顺序依次排列正确的一组是 ( )
A.C、CO、CO2、CaCO3 B.C、CO2、CO、Na2CO3
C.CO、C、CO2、CaCO3 D.CO2、C、CO、CaCO3
查看答案和解析>>
湖北省互联网违法和不良信息举报平台 | 网上有害信息举报专区 | 电信诈骗举报专区 | 涉历史虚无主义有害信息举报专区 | 涉企侵权举报专区
违法和不良信息举报电话:027-86699610 举报邮箱:58377363@163.com