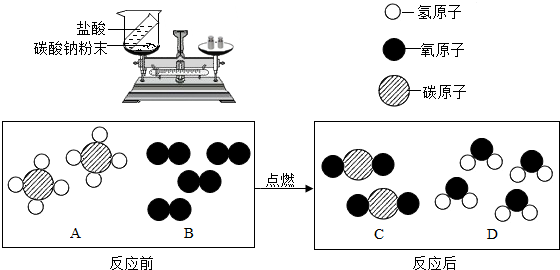
Ϊ�˲ⶨijƷ��ʳ�ô�����̼���Ƶ�����������ijУ��ѧ�о���ѧϰС���̽���������£�
[�������]��Ʒ��̼���Ƶ����������Ƕ��٣�
[֪ʶ��]ʳ�ô������Ҫ�ɷ���̼���ƣ���������������Ȼ��ƣ� ��Ӧ�����в�����ˮ���Ȼ���Ļӷ���
[��Ʒ���]
��1������һ����һ������Ʒ�м����������ʯ��ˮ�����ݷ�Ӧ����̼��Ƶ������������̼���Ƶ��������ټ�����Ʒ��̼���Ƶ�����������
��2������������һ������Ʒ�м���������ϡ���ᣬ���ݷ�Ӧ���ɶ�����̼�������������̼���Ƶ��������ټ�����Ʒ��̼���Ƶ�����������
[����ʵ��]
����ͬѧ����ȡ12.00g��Ʒ����ˮ�����Һ������Һ�м�������ij���ʯ��ˮ�����ˡ�ϴ�ӡ�������õ���ɫ����10.00g��
����ͬѧ����ȡ12.00g��Ʒ������������ϡ����ֱ����Ӧֹͣ�����ռ���4.40g������̼��
[�������]������ѡһ��ͬѧ��ʵ�������������Ǽ������Ʒ��̼���Ƶ���������������������ȷ��0.1% ��
[������˼]
��1�������С��ͬѧ��Ϊ��Ҫ���̼���Ƶ�������Ҳ����ʹ���������ʯ��ˮ�������ͬ����������
����һ�־������ʣ�����Һ����Ʒ��Ӧ��ͨ���ⶨ������ʵ������������йؼ��㼴�ɣ�
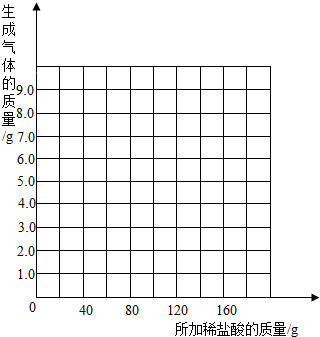
��2�������С��ͬѧ��Ϊ������ϡ�����������������Ҳ�������ȡ18.45g��Ʒ�����ձ��У�ÿ�μ���20gϡ���ᣨ������ˮ���Ȼ����ݳ������þ���������������¼ʵ���������£�
| ����ϡ������� |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
| �ձ�����������������/g |
78.90 |
97.80 |
116.70 |
135.60 |
155.05 |
175.05 |
195.05 |
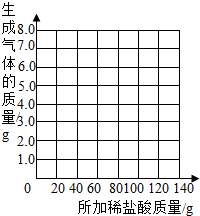
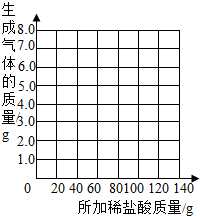
��������ʵ�����ݵı仯���ɣ���ȡ��Ӧ���������������ͨ���йؼ��㣬�������ϡ����������������������⣺�������ұߵ�����ֽ�ϻ����������������������ϡ����������ϵ�����ߣ�

 Ϊ�˲ⶨijƷ��ʳ�ô�����̼���Ƶ�����������ijУ��ѧ�о���ѧϰС���̽���������£�
Ϊ�˲ⶨijƷ��ʳ�ô�����̼���Ƶ�����������ijУ��ѧ�о���ѧϰС���̽���������£�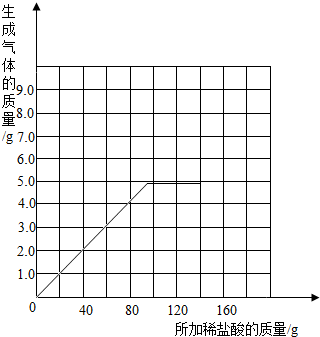


 �¿α�ͬ��ѵ��ϵ�д�
�¿α�ͬ��ѵ��ϵ�д� һ����ʦ����Ӧ����������һ��ȫϵ�д�
һ����ʦ����Ӧ����������һ��ȫϵ�д�