
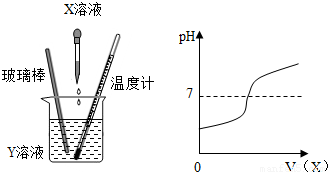
��4�֣���X��Һ����Y��Һ�У��ڵμӹ����У�Y��Һ��pH�仯�����ͼ��ʾ��
| ��Һ | A | B | C | D |
| X | ���� | �������� | ���� | �������� |
| Y | ˮ | ˮ | �������� | ���� |
��1��D δ����X��Һʱ��Y��Һ������
��2����NaOH+HCl=NaCl+H2O
(3)�����ߺ�
��4�����ɺ�����ٱ���
��5����NaCl �����ܼ�
���������������1����ͼʾ��֪����Һ��PH��С��7����Ϊ����7���ʱ����з������ֱ仯�������D��������δ����X��Һʱ��Y��Һ�����ԣ�����X�ĵμӣ���Һ��Ϊ���ԣ�
(2)�÷�Ӧ�Ļ�ѧ����ʽΪNaOH+HCl=NaCl+H2O��
(3)����X��Һ�IJ��ϵ��룬���ձ�����Һ���¶ȱ仯�������ߺͣ���Ϊ������������Ʒ�Ӧ�ų������������������ƹ�������Һ���¶Ƚ����ͣ�
(4)��Y��Һ�е���ʯ���Լ��������ϵ���X��Һ��ʯ��ı仯���ɺ�����ٱ�����
(5)��X��Yǡ����ȫ��Ӧʱ����Һ�е�������NaCl����Ҫ������Һ�л�ô������壬Ӧ�ò��������ܼ��ķ�����
���㣺��Һ��PH�����ָʾ������ѧ��Ӧ�е������仯��������
������PH����7����Һ�Ǽ��Եģ�С��7�������Եģ�����7�������Եġ�
��ɫʯ����Һ�����ָʾ���������������ʱ��ɫ�������������ʱ���ɫ��
�Ȼ��Ƶ��ܽ�������¶ȱ仯�����ӣ���������Ȳ���һ����������ܼ��ķ������Ȼ�����Һ�еõ��Ȼ��ƾ��塣



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��X��Һ����Y��Һ�У��ڵμӹ����У�Y��Һ
��X��Һ����Y��Һ�У��ڵμӹ����У�Y��Һ| ��Һ | A | B | C | D |
| X | ���� | �������� | ���� | �������� |
| Y | ˮ | ˮ | �������� | ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��X��Һ����Y��Һ�У��ڵμӹ����У�Y��Һ��pHֵ�仯�����ͼ��ʾ�����ƶ�X�Ļ�ѧʽΪ
��X��Һ����Y��Һ�У��ڵμӹ����У�Y��Һ��pHֵ�仯�����ͼ��ʾ�����ƶ�X�Ļ�ѧʽΪ| ��Һ | A | B | C | D |
| X | ���� | ���� | �������� | �������� |
| Y | ˮ | �������� | ���� | ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 ��X��Һ����Y��Һ�У��ڵμӹ����У�Y��Һ
��X��Һ����Y��Һ�У��ڵμӹ����У�Y��Һ| ��Һ | A | B | C | D |
| X | ���� | �������� | ���� | �������� |
| Y | ˮ | ˮ | �������� | ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�����ʡ�˲���ѧ���꼶���£���һ���¿���ѧ�Ծ��������棩 ���ͣ������
| ��Һ | A | B | C | D |
| X | ���� | �������� | ���� | �������� |
| Y | ˮ | ˮ | �������� | ���� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com