
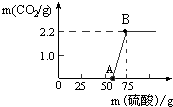
 ʵ������һƿ���õ�NaOH����ȡ13.3g ��NaOH��Ʒ������ΪNa2CO3�����ձ��У�Ȼ�����ձ�����μ�����������19.6%��ϡ���ᣬ��Ӧ�����ͼ��ʾ��
ʵ������һƿ���õ�NaOH����ȡ13.3g ��NaOH��Ʒ������ΪNa2CO3�����ձ��У�Ȼ�����ձ�����μ�����������19.6%��ϡ���ᣬ��Ӧ�����ͼ��ʾ��| 106 |
| x |
| 44 |
| 2.2g |
| 98 |
| 75��19.6% |
| 142 |
| y |
| 21.3g |
| 86.1g |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ijУ�о���ѧϰС�������һ����Ȥ��ʵ��̽����
ijУ�о���ѧϰС�������һ����Ȥ��ʵ��̽�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
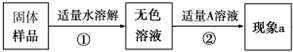 ʵ������һƿ���õ��������ƹ��壬ijͬѧ���˽����Ƿ���ʣ����������ʵ�飬������һ���������̽�����
ʵ������һƿ���õ��������ƹ��壬ijͬѧ���˽����Ƿ���ʣ����������ʵ�飬������һ���������̽������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
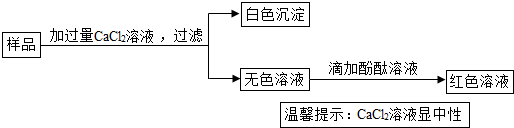
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ����ˮ�����Һ |
| ���������CaCl2��Һ |
| ���ˡ�ϴ�� |
| ������� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com