
| С���ϣ� �Ȼ�����Һ��PHΪ7�� NaCO3+CaCl2=2NaCl+CaCO3�� 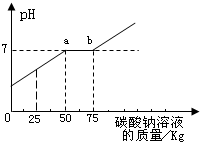 ��1��ͨ��ͼ������Ͱ�еķ�Һ�ﺬ�е������� ��2��ͨ��ͼ��֪����̼������Һ�����ӵ� ��3��ͼ�б�ʾa��ĺ��� ��4������������Һ���Ȼ��Ƶ�����Ϊ ������̼�����ϡ���ᷴӦ�����Ȼ�������ˮ������ͼ���е�PH�ж���Һ�е����ʣ�����̼���Ʒ�����Ӧ���Ⱥ�˳���ٽ��ͼ����м��㣮 ����⣺��1����ͼ��ɿ�������Ӧ��ķ�Һ��pHС��7����������ʣ�࣬���Ͱ�з�Һ������ΪHCl�����ɵ�CaCl2�� ��2���ڷ�Һ�м���̼���ƣ��������ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��Ȼ��̼�������Ȼ��Ʒ�Ӧ����̼��Ƴ�������Ϊ��ʹ�����Ȼ��Ʒ�Ӧ����̼��ƣ�����̼��ƾͱ������ܽ��ˣ�����̼���ƵIJ��ϵμӣ�PH�������ߣ�������ǡ����ȫ��Ӧ��ʱpH����7��֮��̼�������Ȼ��Ʒ�Ӧ��pH���䣬���Ȼ��Ʒ�Ӧ�̼꣬������ʣ��ʱpH����7����ͼ������ηֱ��ʾ̼���������ᷴӦ��̼�������Ȼ��Ʒ�Ӧ��̼����ʣ�࣮��ͼ�����ݿ�֪��̼������Һ�����ӵ�75kgʱ����Һǡ�ô����꣮ ��3��ͼ�б�ʾa��ĺ���a����̼����������ǡ����ȫ��Ӧ��pH����7����Һ�����ԣ� ��4����������д����ѧ����ʽNa2CO3+2HCl�T2NaCl+H2O+CO2����Na2CO3+CaCl2=CaCO3��+2NaCl���ݷ�Ӧǰ��Ԫ����������ɿ����μӷ�Ӧ��̼�����е���Ԫ��ȫ���������Ȼ��ƣ����ҳ�̼�������Ȼ��ƵĹ�ϵʽ�����ݹ�ϵʽ������Ȼ��Ƶ������� ���Ȼ��Ƶ�����Ϊx �ɻ�ѧ����ʽ Na2CO3+2HCl�T2NaCl+H2O+CO2����Na2CO3+CaCl2=CaCO3��+2NaCl�� �ɵù�ϵʽ��Na2CO3��2NaCl 106 117 75kg��21.2% x
x=17.55kg �ʴ�Ϊ�� ��1��HCl��CaCl2 �� ��2��75 ��3��a����̼����������ǡ����ȫ��Ӧ��pH����7����Һ�����ԣ� ��4��11.7 kg+5.85kg=17.6 kg�� ����������ؽ��ǽ���������ͼ������α�ʾ�����壮����Ԫ�������غ㷨����ȸ��ݻ�ѧ����ʽ����  
��ϰ��ϵ�д�
 ������������Ӧ����ϵ�д� ������������Ӧ����ϵ�д� ͬ����չ�Ķ�ϵ�д� ͬ����չ�Ķ�ϵ�д�
���ϰ��
��Ŀ�����л�ѧ ��Դ�� ���ͣ� 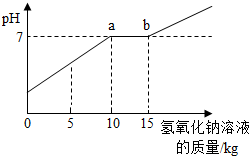 ��2009?���ţ�ͨ����ʵ�������ꡰ��������ȡ��ʵ���Ҫ����Һ�����ҺͰ�У������Ϳ��ռ�һЩ���������п�Ļ����Һ���������������ʣ���Ϊ������Ⱦ������ijУ��ѧ��ȤС���ͬѧ��������ʵ�飺 ��2009?���ţ�ͨ����ʵ�������ꡰ��������ȡ��ʵ���Ҫ����Һ�����ҺͰ�У������Ϳ��ռ�һЩ���������п�Ļ����Һ���������������ʣ���Ϊ������Ⱦ������ijУ��ѧ��ȤС���ͬѧ��������ʵ�飺ȡ��ҺͰ�ϲ���Һ��12.2kg�������м���������������Ϊ8%������������Һ��������ҺpH����������������Һ��������ϵ��ͼ��ʾ������֪ZnSO4+2NaOH�TZn��OH��2��+Na2SO4��ZnSO4��Һ��pH�ӽ�7�� ��1��ͼ�б�ʾ��a��ĺ����ǣ� ��ʾ��������������ǡ����ȫ��Ӧ ��ʾ��������������ǡ����ȫ��Ӧ ����2��ͨ��ͼ��֪��������������Һ�������ӵ� 15 15 kgʱ����Һǡ�ô����꣮�鿴�𰸺ͽ���>> ��Ŀ�����л�ѧ ��Դ�� ���ͣ������ ������ʵ�������ꡰ������̼����ȡ��ʵ�����Һ����Ͱ���ռ��˴����Ļ����Һ�����ǹ������ʣ���Ϊ������Ⱦ��������ѧ��ȤС����������ʵ�飺
�鿴�𰸺ͽ���>> ��Ŀ�����л�ѧ ��Դ����ƽ��һģ ���ͣ������ ������ʵ�������ꡰ������̼����ȡ��ʵ�����Һ����Ͱ���ռ��˴����Ļ����Һ�����ǹ������ʣ���Ϊ������Ⱦ��������ѧ��ȤС����������ʵ�飺 ȡ��ҺͰ�ϲ���Һ��59.4kg�������м���������������Ϊ21.2%��̼������Һ��������ҺpH������̼������Һ��������ϵ��ͼ��ʾ��
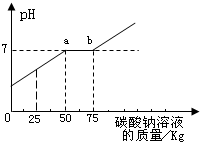 ��1��ͨ��ͼ������Ͱ�еķ�Һ�ﺬ�е�������______�� ��2��ͨ��ͼ��֪����̼������Һ�����ӵ�______kgʱ����Һǡ�ô����꣮ ��3��ͼ�б�ʾa��ĺ���______�� ��4������������Һ���Ȼ��Ƶ�����Ϊ______�� �鿴�𰸺ͽ���>> ��Ŀ�����л�ѧ ��Դ��2009�걱���в�ƽ���п���ѧһģ�Ծ��������棩 ���ͣ������ ��2009?��ƽ��һģ��������ʵ�������ꡰ������̼����ȡ��ʵ�����Һ����Ͱ���ռ��˴����Ļ����Һ�����ǹ������ʣ���Ϊ������Ⱦ��������ѧ��ȤС����������ʵ�飺 ȡ��ҺͰ�ϲ���Һ��59.4kg�������м���������������Ϊ21.2%��̼������Һ��������ҺpH������̼������Һ��������ϵ��ͼ��ʾ��
 ��1��ͨ��ͼ������Ͱ�еķ�Һ�ﺬ�е������ǣ� ��2��ͨ��ͼ��֪����̼������Һ�����ӵ�kgʱ����Һǡ�ô����꣮ ��3��ͼ�б�ʾa��ĺ��壮 ��4������������Һ���Ȼ��Ƶ�����Ϊ�� �鿴�𰸺ͽ���>> ͬ����ϰ��� ����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר�� Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com��Ȩ��������վ�������£�ͼƬ��Դ�����磬����Ȩ����Ȩ��ԭ�������У�ת�������ַ���Ȩ��������Ȩ����������������֪�����ǽ����촦������ϵqq��3310059649�� ICP�������: ��ICP��07509807��-10 ����������42018502000812�� | |||||||||||||||||||||||||||