
ij�����Ʒ�к����Ȼ������ʡ�Ϊ�ⶨ��Ʒ��̼���Ƶ�������������20��ʱ����ȡ����Ʒ26.5g�����뵽ʢ��һ������ϡ������ձ��У�̼������ϡ����ǡ����ȫ��Ӧ��������ȫ�ݳ����õ�������NaCl��Һ����Ӧ�����þ�����������ձ��ڻ�����������m���뷴Ӧʱ�䣨t����ϵ��ͼ����
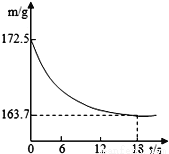
��1������CO2������__________��
��2���ô�����Ʒ��Na2CO3����������__________��
��3����ȫ��Ӧ��������Һ��������������___________������������ȷ��0.1%��
8.8g 80% 17.5% ����������1�����������غ㶨�ɣ���Ӧǰ�����ʵ����������䣬���ɶ�����̼������Ϊ172.5g-163.7g=8.8g����̼���Ƶ�����Ϊx���Ȼ��Ƶ�����Ϊy Na2CO3+2HCl = 2NaCl+H2O+CO2�� 106 117 44 x y 8.8g x=21.2g y =23.4g ���... ѧϰʵ����ϵ�д�
ѧϰʵ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�������к�����2018����꼶�ڶ�ѧ��������ϰ ���ͣ���ѡ��
�������������Լռ21%���ǣ� ��
A. ���� B. ���� C. ������̼ D. ϡ������
B ����������������������ɷ��У��������Լռ21%�����������ʴ�ѡ��B�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�������а�����2018����꼶�п�һģ��ѧ�Ծ� ���ͣ���ѡ��
���������������������
A. Һ�� B. Cl2O7
C. CH3COOH D. FeSO4��7H2O
B ����������������Ԫ�صĻ��������һ��Ԫ������Ԫ�أ������Ļ������������A. Һ�����ڵ��ʣ�����B. Cl2O7�����������ȷ��C. CH3COOH�����ᣬ����D. FeSO4��7H2O�����Σ�����ѡB���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ������2017����꼶ȫ���п�ģ�⻯ѧ�Ծ� ���ͣ������
�˵����Ϊ1��18��Ԫ�ص�ԭ�ӽṹʾ��ͼ����Ϣ���£��ش��������⣺

(1)�ڵ��������У���ԭ�ӽṹ�Ĺ�֮ͬ���� ________________________���������У���ԭ�Ӻ�������Ų��ı仯������__________________________ ��
(2)�ڵ��������У�Ԫ�����͵ı仯����ǣ��������� ______Ԫ�ع��ɵ�_____Ԫ�أ�����ϡ������Ԫ�ؽ�β��
(3)д����������Ų�����ԭ����ͬ�������Ӻ������ӷ��Ÿ�һ����������____________��������_________��
ԭ�Ӻ�����Ӳ�����ͬ ������������������ ���� �ǽ��� Na+(Mg2+��Al3+) O2-(��F-) ��������(1)�ڵ������ڣ����У���ԭ�ӽṹʾ��ͼ��֪����ԭ�ӽṹ�Ĺ�֮ͬ���ǵ��Ӳ�����ͬ���������У���ԭ�Ӻ�������Ų��ı仯�����������������������ࣻ(2)�ڵ��������У�Ԫ�����͵ı仯����ǣ��������ɽ���Ԫ�ع��ɵ��ǽ���Ԫ�أ�����ϡ������Ԫ�ؽ�����(3)��ԭ�Ӻ����������10������һ...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ������2017����꼶ȫ���п�ģ�⻯ѧ�Ծ� ���ͣ���ѡ��
�ס������ֹ������ʵ��ܽ��������ͼ��ʾ�������й������д������
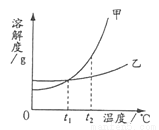
A. t2��ʱ���ܽ�ȱ��ҵĴ�
B. ���ס��ҵı�����Һ��t2�潵��t1�棬������������
C. �����¶ȿɽ��ı�����Һ��ɲ�������Һ
D. t1��ʱ�����ҵı�����Һ��100g�������ʵ�����һ�����
B �������������������ͼt2����ܽ�ȱ��ҵĴ�A��ȷ�����������ס��ҵı�����Һ��t2����t1��������������B�����ܽ�����¶����߶����������¶ȿ��Խ��ı�����Һ��ɲ�������ҺC��ȷ��t1��ʱ���������ʵ��ܽ����ȣ����ҵı�����Һ��100g�������ʵ�����һ����ȣ�D��ȷ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���Ĵ�ʡ������������2018����꼶��ѧ�ڶ��ﻯѧ�Ծ� ���ͣ���ѡ��
�۲�ʵ��ͼ�õ�������ȷ����

A. ͼһ����һ����������������Һ�в��ϵμ�ˮ
B. ͼ������ʯ��ˮ�м���������CaO
C. ͼ������100 g Cu-Zn�Ͻ��м����������ᣬ��������������֪Zn����������Ϊ50%
D. ͼ�ģ������������ͭ�Ļ����Һ�еμ�����������Һ
B ��������A.��һ����������������Һ�в��ϵμ�ˮ����Һ�ļ��Ա�����pH��С����������Һʼ�ճʼ��ԣ�pH������ڻ�С��7������B.�������Ƶ��ܽ�����¶����߶���С����ʯ��ˮ�м���������CaO�������ƺ�ˮ��Ӧ�ų��������ȣ��ʷ�Ӧ���������Ƶ��ܽ�ȱ�С����Ӧ��������Һ���¶���������ʼ�¶ȡ������£��˹������������Ƶ��ܽ�ȱ����ȷ��C.��100g Cu-Zn�Ͻ��м����������ᣬ��������...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ�ʮУ2018����꼶��ѧ��������ѧ�Ծ� ���ͣ�������
��ҵ�ϴ���40t������ͭ����ͭ����ˮ��������8t��5%�������Ƶ���ֽ����ˮ����ش��������⣺
��1��8t��ֽ����ˮ�к��е�������������Ϊ______________��
��2��������ͭ����ˮ������ͭ����������______________��
0.4t 2% ����������1������������Ϣ�����𣻣�2�����ݷ�Ӧ�Ļ�ѧ����ʽ�����𡣣�1��8t��ֽ����ˮ�к��е�������������Ϊ8t��5��=0.4t����2������ͭ����ˮ������ͭ������Ϊx�� 2NaOH+CuSO4=Na2SO4+Cu(OH) 2�� 80 160 0.4t x x=0.8t ��ͭ����ˮ������ͭ����������Ϊ��100��=2% �𣺣�1��8t��ֽ...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ɽ��ʡ̩���������2017-2018ѧ����꼶��ѧ�����п��Ի�ѧ�Ծ� ���ͣ������
�ҹ����������ġ��������������������±�������PM2.5���ָ�ꡣPM2.5��ָ������ֱ��С�ڻ����2.5�Ŀ������Ҫ���Ի�ʯȼ�ϵ�ȼ�պ��ﳾ�������彡���Ϳ���������Ӱ��ܴ�
(1)���������Ⱦָ������Ŀ�г��˿�����������⣬������ ___________ (�����)��
��CO����CO2����SO2����NO2
(2)ij�дӽ���4��1�������л�������Ҫ����β����⣬���а���PM2.5�ļ�⡣����ȼ�ղ����Ŀ�������Ҫ��____________���������Ͳ���ȫȼ�ղ����ġ�
(3)���й���PM2.5��˵���������____(�����)��
��ȼ���̻���������PM2.5����PM2.5��������������ġ�Ԫ�ס�֮һ����PM2.5�Ŀ�����С����Ӵ�С��ࡡ��PM2.5ר�ÿ���ʹ���˻���̿���������˻���̿��������
(4)���˶�����β���ļ�⣬������Щ��ʩ��PM2.5���������������ã����һ����_______________________��
�٢ۢ� ̼(��̼������̼��) �� ���ٻ������磬�ᳫʹ��ˮ�ܡ�̫���ܷ��磻�����ֲ�����֣������ﳾ��Ⱦ(�Ӽ��ٻ�ʯȼ�ϵ�ȼ�գ�ʹ�������Դ��ֲ�����ֿ����� ����������1��Ŀǰ���������Ⱦָ�����к�������̳���Ҫ��������������һ����̼����������������������������������������ȣ�����Ӧ��٢ۢܣ� ��2�����Ͳ���ȫȼ�ջ����������̿�ڣ����Dz���ȫȼ�ղ�����̼�� ��3��PM2.5��...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ2018����꼶�ڶ���ģ�⿼�Ի�ѧ�Ծ� ���ͣ���ѡ��
������ϴ�����У������黯���õ���
A. ������ˮϴ�� B. ��������ϴ����
C. ��ϴ�Ӽ���ϴ����IJ;� D. ��ʳ�����ˮ��
C ��������A��������ˮϴ��û�������黯���ã�����B��������ϴ�������õ������Ͷ����۵��ܽ����ã�����C����ϴ�Ӽ���ϴ����IJ;����õ���ϴ�Ӽ������۵��黯���ã���ȷ��D����ʳ����ϴ���ˮ�������ô�������ˮ����Ӧ�����ʣ�û�������黯���ã�����ѡC���鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com