
��8�֣������и����е�������Һ��ϣ���Һ�е�ԭ�е�ˮ���Ӻ��Բ�д����
| ��� | ��Ӧ��1 | ��Ӧ��2 | ��Ϻ���ڵ����� |
| A | ϡ���ᣨH+��Cl-�� | ����������Һ(K+��OH-) | K+��Cl-��H2O |
| B | | | Na+��SO42-��Cu(OH)2 |
| C | �Ȼ�����Һ(Na+��Cl-) | ϡ���ᣨH+��SO42-�� | H+��SO42-��Na+��Cl- |
��1��HCl + KOH = KCl + H2O H+ OH- H2O
��2������ͭ��Һ ����������Һ
��3�����ǰ����Һ�е�����û�н�ϳ��µķ���
���������������1��A����ϡ���������������Һ���Ӧ����ѧ����ʽΪ��HCl + KOH = KCl + H2O������ͼ�����кͷ�Ӧ���кͷ�Ӧ��ʵ���ǣ�����H+�ĺͼ���OH-�������γ�ˮ
��2����Ϊ��Ӧ����ڵ�����Ϊ��Na+��SO42-��Cu(OH)2�����ҷ�Ӧ��1���ε�ˮ��Һ����Ӧ��2�Ǽ��ˮ��Һ�����Կ��Ƴ���Ӧ��1������ͭ��Һ����Ӧ��2������������Һ����Ӧ����ʽΪ��CuSO4+2NaOH==Cu(OH)2��+Na2SO4
��3�����ֽⷴӦ�����������ǣ����������г����������ˮ���ɣ�C��û�з�����ѧ��Ӧ��ԭ����(�۽ǶȽ���)�����ǰ����Һ�е�����û�н�ϳ��µķ���
���㣺���ֽⷴӦ����������


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ѧ������ϢϢ��أ���������ѧ��ѧ֪ʶ�ش����������е����⡣
��1��С����ѧһ�����ž��ŵ����㣬��֤���������� ���ʡ�
��2��С������������ϴ�·���ˮ�м������ˮ�����������״���������Ϊ��ʱ��ˮΪ ���Ӳˮ������ˮ������
��3������ˮ����ˮ�����г��û���̿������̿����Ҫ������ ��
��4��ϴ�Ӽ�����ϴ�;��ϵ����ۣ�������Ϊϴ�Ӽ����� ���ܡ�
��5����ʯ�ҳ�����ʳƷ���������ԭ���ǣ��û�ѧ����ʽ��ʾ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��4�֣�ij��ѧ��ȤС���ͬѧ����ͼ��ʾװ�ý���ʵ�飨װ�����������ã���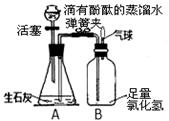
��1���ӷ�Һ©������һ����Һ�壬�����رջ��������ɼУ�A���ܹ۲쵽�������� ��
��2��δ�ָ������£����رյ��ɼУ�Bƿ�в��������ԭ����
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��8�֣�
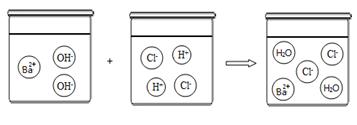
��1�����й��������������ʵ������У����ڼ�Ĺ�ͬ���ʵ���
| A����ʹ��ɫʯ����Һ����ɫ | B���������ᷴӦ����ˮ |
| C������Na2SO4��Һ��Ӧ����BaSO4���� | D������CO2��Ӧ����ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��5�֣�����������������Һ������Ӧ��������Һ��pH�仯��ͼһ����������ش����⣺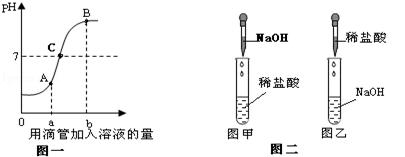
��1����ͼһ���жϣ��÷�Ӧ��ʵ������ǰ���ͼ���е� ���ͼ�ס���ͼ�ҡ��������еġ�
��2��ͼһ��A��B��C�����ʾ����Һ�����ַ�Ӧ��ǡ����ȫ��Ӧ�ĵ��� �㣬������ɫʯ����Һ����ֺ�ɫ���� �����Һ����ʱ��Һ�еĺ��е��������� ��
��3������̽������������������Һ��Ӧ��������С����ʵ��������£�
ʵ����̣��Ȱ�ͼ�ײ������������������Һ���۲������ְ�ɫ�������ó����ۣ�֤���������Ȼ��ơ�С����Ϊ������۲���ȷ�������Ľ����� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������������ѡ����գ�����ţ�
| A������ | B���ƾ� | C����ʯ�� | D��ͭ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��4�֣��±��Ǽ��ּ����������й���Ϣ��
| �������� | ����� | ������Ư | Ư�� |
| ��Ч�ɷ� | HCl | H2O2 | NaClO |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��9�֣��ڡ����뻯ѧ����У�ij��ȤС���ͬѧ��ʵ�����ú���������ʯ�ҵ�NaCl��������һ������������NaCl��Һ��ʵ�鲽������ͼ��ʾ��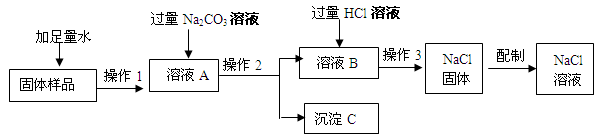
��ش�
��1���ڡ�������Ʒ���м���ˮʱ��������Ӧ�Ļ�ѧ����ʽΪ ��
��2������ҺA������������ ���ѧʽ�������롰����Na2CO3����������Ӧ�Ļ�ѧ����ʽ ��
��3���ڡ�����2���У�Ҫ�õ��IJ������������������ձ��⣬���� ��
��4�����롰����HCl��Һ����Ŀ���� ��
��5��������3���������� ��
��6����Ҫ����100g��������Ϊ10%���Ȼ�����Һ����Ҫˮ������ g����Ҫ�õ����������⣬����Ҫ�IJ��������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��4 �֣����� 60 ���������ҹ���ѧ������ѧ�ķ�չ��ȡ���˾�����Ŀ�ijɾ͡��ҹ���ѧ������ֽ�Ͷ������ѣ�TiO2����Ĥ������һ�����͡�����ֽ������������ֽ�ϡ��̡�һ�㡰��������C10H9N��Ⱦ�ϣ��Ƴ�һ����ֽ�����ڼ��ʳƷ����������Ũ�ȵĸߵ͡�
��1������������ Ti �Ļ��ϼ�Ϊ ��
��2��������һ�������У�C��H��N ����ԭ�ӵĸ���֮��Ϊ ������������ȣ���
��3����֪ NaNO2 �ܷ������·�Ӧ����ѧ����ʽ����ƽ����
2NaNO2+2KI+2H2SO4=I2+2M��+2H2O+Na2SO4+K2SO4���� M �Ļ�ѧʽ�� ��
��4������������Ӧ�����û�ѧ��ֽ�������г������ʽ���ʵ���������������κ�ʳ�Σ����е⻯�ص�����ֽ������ѡ�����������еij�������Ϊ________������ţ���
��ʳ���� �ڰ� ��ʳ�� ��ţ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com