ijУ��ѧ�о���ѧϰС���ͬѧ��һƿ���ڷ��õ��������Ʋ�������Ȥ����������ͬѧΧ����ƿ�������ƽ�����һϵ�е�̽�����
�������
�������
�����������Ƿ���ȫ���ʢ���Ʒ��̼���Ƶ����������Ƕ��٣�
��������
��������
�����������������̼��Ӧ����̼���ƺ�ˮ��
�����������ƺ�̼���ƻ�����м�������ʱ�����������������Ʒ�Ӧ������̼���Ʒ�Ӧ��
���ʵ��
���ʵ��
ʵ��һ��̽���������ƹ���ı��������
��ȡ�����ù�����Ʒ�����Թ��У������м���ϡ���ᣬ���������ݲ�����˵������Ʒ�к���̼���ƣ��ɴ˿�ȷ���ù����ѷ������ʣ���Ӧ����ʽΪ��
Na2CO3+2HCl=2NaCl+H2O+CO2��
Na2CO3+2HCl=2NaCl+H2O+CO2��
��
��������������Ѿ���ȫ���ʣ�����ͬѧ��Ϊȡ������������ˮ�������еμ���ɫ��̪����ҺӦ����ɫ������ͬѧ˼������Ϊ����ͬѧ���뷨�Ǵ���ģ�ԭ���ǣ�
̼���ƺ�����������Һ���Լ��ԣ�ʹ��̪��Һ��ɺ�ɫ
̼���ƺ�����������Һ���Լ��ԣ�ʹ��̪��Һ��ɺ�ɫ
��
��Ϊ̽���ù������Ƿ���δ���ʵ��������ƣ�ͬѧ���ֽ��������±���ʾ��ʵ�飮�뽫�±���д������
| ʵ��Ŀ�� |
ʵ����� |
���� |
���ۻ�ѧ����ʽ |
| ��ȥ̼���� |
ȡ�����ù�����Ʒ����ˮ�����Һ���μ��������Ȼ�����Һ����ַ�Ӧ����� |
������ɫ���� ������ɫ���� |
�йط�Ӧ�Ļ�ѧ����ʽΪ
Na2CO3+CaCl2=2NaCl+CaCO3�� Na2CO3+CaCl2=2NaCl+CaCO3�� |
| �����Ƿ����������� |
����˺����Һ�е��� ��̪��Һ ��̪��Һ |
��� ��� |
����Ʒ�к����������� |
ʵ������ⶨ����̼���Ƶ���������
��Ʒ���
��Ʒ���
�ټ���ͬѧ����ȡ20.00��Ʒ���������������ֱ����Ӧֹͣ�����ռ���4.40g������̼�������˼·��������Ʒ�����ᷴӦ���ɶ�����̼���������̼���Ƶ��������ټ�����Ʒ��̼���Ƶ�������������
������ͬѧ����ȡ20.00g��Ʒ����ˮ�����Һ������Һ�м�������ij���ʯ��ˮ�����ˡ�ϴ�ӡ�������õ���ɫ����10.00g�������˼·��������Ʒ��ʯ��ˮ��Ӧ���ɳ���̼��Ƶ������������̼���Ƶ��������ټ�����Ʒ��̼���Ƶ�������������
�������
�������
��������ͬѧ��ʵ����������������Ǽ������Ʒ��̼���Ƶ���������������ѡһ��д��������̣�
�����뷴˼
�����뷴˼
��1���ڽ���ʱ����ʦ��ͬѧ�ǡ��ܷ�������ʵ�����õ����ݼ������Ʒ���������Ƶ����������������������ͬѧ��һ����Ϊ���ԣ���С��˼�����üס������ַ��������ף������ǣ�
��Ʒ����ˮ���������Ʒ���������Ƶ�����
��Ʒ����ˮ���������Ʒ���������Ƶ�����
��
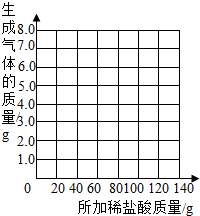
��2��С������������·��������õ��ӳ�ȷ��ȡ20.00g���ʵ�NaOH��Ʒ������ƿ�У��õ��ӳӳӵ���ƿ����Ʒ��������Ϊ70.00g���ٰ�175.00g7.3%ϡ����ƽ���ֳ�7�����μ�����Ʒ�У�ÿ�γ�ַ�Ӧ�õ��ӳӳӵ���ƿ����ʢ���ʵ�������ʵ�����ݼ�¼���£�
| ��������Ĵ��� |
��һ�� |
�ڶ��� |
������ |
���Ĵ� |
����� |
������ |
���ߴ� |
| ��ƿ����ʢ���ʵ������� |
95.00 |
120 |
145 |
170 |
192.8 |
215.6 |
240.6 |
��2�����������ʵ�����ݣ�������Ʒ���������Ƶ���������Ϊ
40.0%
40.0%
����Ҫд��������̣�




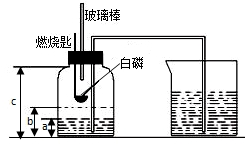 ijУ��ѧ�о���ѧϰС����ѧϰ�ˡ����������������ⶨ���Ļ����ϣ��Ľ��˽̲��е�ʵ�飬��Ƴ�����ͼ��ʾ��ʵ��װ�ã�ʵ�鲽�����£�
ijУ��ѧ�о���ѧϰС����ѧϰ�ˡ����������������ⶨ���Ļ����ϣ��Ľ��˽̲��е�ʵ�飬��Ƴ�����ͼ��ʾ��ʵ��װ�ã�ʵ�鲽�����£�