
����Ŀ���±���Ca��OH��2��NaOH���ܽ�����ݣ���ش��������⣺
![]()
��1�������ϱ����ݣ�����Ca��OH��2��NaOH���ܽ�����ߣ���ͼ���ܱ�ʾNaOH�ܽ�����ߵ��� ����A��B����
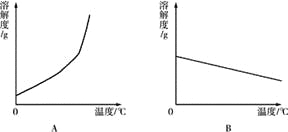
��2��Ҫ���һƿ�ӽ����͵�Ca��OH��2��Һ��ɱ�����Һ�������ʩ�У�
�������������ƣ��������¶��������¶ȣ�������ˮ��������ˮ���ٻָ���ԭ�¶ȣ���������ʯ�ң�
���д�ʩ��ȷ���ǣ� ��
A���ڢܢ� B���ۢ� C���٢ۢݢ� D���٢ڢݢ�
��3��20��ʱ��191 g����NaOH��Һ������10 gˮ���ٽ��µ�20����������NaOH���������Ϊ ��
��4������20��ʱCa��OH��2�ı�����Һ������Һ���������м���һ����CaO��õ�����Һ������Һ������ʱ��Һ�����ʵ����������� �ף�����������������������������
��5������60��ʱ��Ca��OH��2��NaOH�������ʵı�����Һ����Ҫ�õ��ϴ�����NaOH���壬Ӧ��ȡ������������ ��
��6��20��ʱ�����ⶨNaOH��Һ��pH�����Ƚ�pH��ֽ������ˮ��ʪ���ٽ��вⶨ����������Һ��pH ������ƫ��������ƫС����������Ӱ��������
���𰸡���1��A����2��D����3��9.1g����4��������5�����½ᾧ�����ˣ���6��ƫС
������������������������Ƶ��ܽ��������¶ȵ����߶���С��Ҫ���һƿ�ӽ����͵�Ca��OH��2��Һ��ɱ�����Һ�������ʩ�У������¶ȡ�����ˮ���ٻָ���ԭ�¶ȡ������������ƣ�20��ʱ��191 g����NaOH��Һ������������91g��ˮ100g��ÿ10gˮ�ܽ�9.1g�������ƣ�����10 gˮ���ٽ��µ�20����������NaOH���������Ϊ9.1g;�������Ƶ��ܽ��������¶ȵ����߶���С���������Ƶ��ܽ��������¶ȵ����߶�����Ca��OH��2��NaOH�������ʵı�����Һ����Ҫ�õ��ϴ�����NaOH���壬Ӧ��ȡ�����������ǽ��½ᾧ����pH��ֽ������ˮ��ʪ����ϡ������������Һ����С������pH��С��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������й�ʵ������������У���ȷ���ǣ�������
A. ������������ȼ��ʱ�����ɴ�����ɫ����
B. ˮͨ������������������
C. ̼��������ȼ��ʱ�������⣬���ɶ�����̼����
D. ��ɫ��̼���ƹ����ϡ�����ϣ��д�����ɫ����ð��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��A��B��C��D����Ԫ�����ڱ���ԭ������Ϊ1-18֮��ij���Ԫ�ء�����Aԭ������һ�����Ӻ�һ�����ӹ��ɵģ�BԪ����ɵĵ����ǿ����к����������ʣ�C2-��D3+�Ľṹʾ��ͼ���ɱ�ʾΪ![]() ���ɴ���ش��������⣺
���ɴ���ش��������⣺
(1)д������Ԫ�ط��ţ�A__________B____________��
(2)C2-������������__________ ,Dԭ�ӵĽṹʾ��ͼ�ɱ�ʾΪ_______________��
(3)д��AԪ����CԪ����ɵ�һ�ִ�����Ļ�ѧ����_________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������ݻ���������(��ƿ�������ܡ�ȼ�ճ�ƿ��������ϸɰ)������װ�������ļ���ƿ������ƽ�������̣�������ƽ�⣬Ȼ��ͬʱѸ�ٷ����ȼ�ĵ������İ���ľ̿(����ͼ��ʾ)ʹ���߳��ȼ�պ���ȴ�����£���ֹˮ�к�ʱ����ƽ
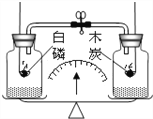
A. �Դ���ƽ��״̬ B. ָ�������������ƫ
C. ָ��ƫ���� D. ָ��ƫ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ij��ɫ��ĩ���ܺ���̼���ơ������ơ�����ء��Ȼ��ء����������е�һ�ֻ��֣�Ϊ��̽����ɷ֣�С��ͬѧ������ͼ��ʾ���̽�����ʵ�飺
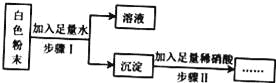
���ڸ�ʵ��������˵����
���������г���ȫ���ܽ⣮��ԭ��ɫ��ĩһ��������������̼���ƣ��������Ȼ���
���������г��������ܽ⣬��ԭ��ɫ��ĩһ��������������̼���ơ������
���������г������ܽ⣮��ԭ��ɫ��ĩһ��������������һ��û��̼����
���������г��������ܽ⣬��ԭ��ɫ��ĩ����ɿ�����3�����
����˵������ȷ�ĸ����ǣ� ��
A. 1�� B. 2�� C. 3�� D. 4��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������йش�����˵���д�����ǣ� ��
A���������Ƕ�������
B�������ܸı��������ʵĻ�ѧ��Ӧ�ٶ�
C���ڻ�ѧ��Ӧǰ�����������û�иı�
D���ڻ�ѧ��Ӧǰ������Ļ�ѧ����û�иı�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��Ϊ�ⶨijBaCl2��Һ������������������������ʵ�飺ȡ104g����Һ���ձ��У�����1gϡ������Һ�ữ�����κ�����Ȼ��30gһ����������������������Һ�����μ��룬ÿ�γ�ַ�Ӧ����ˡ�������ʵ�������ʵ�������ʧ���Բ��ƣ������������
��1�� | ��2�� | ��3�� | |
����������Һ������/g | 10 | 10 | 10 |
���˺���Һ������/g | 105 | 105 | 111.7 |
�����������ݼ��㣺��BaCl2+H2SO4�TBaSO4��+2HCl��
��1����ʵ������в���������������Ϊ g��
��2����BaCl2��Һ�����ʵ�����������д��������̣���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������������ʹ���ж����ǣ� ��
A���ù�ҵ�ƾ����ư�����
B����ȼ����ˮ����װ��ԡ����
C�����߲˴�����ͨ��������CO2
D���õ���������ˮ��������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����һ���¶��£���20g Na2CO3��NaCl�Ĺ��������м���101gˮ���ܽ���������м���100g CaCl2��Һ��ǡ����ȫ��Ӧ�����ˣ�������Һ������Ϊ211g��������ϴ�ӡ�����Ƶ�������Ϊm���Լ��㣺
(1)m����g��
(2)��ǡ����ȫ��Ӧʱ��������Һ�����ʵ���������Ϊ���٣�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com