��ϰʱ������ר��̽���ķ���ѧϰ�������ʵ���ɡ���������ȡ����������ʦ�������ǻع��˼���̽��ʵ�飮
��1��̽��1���о������������
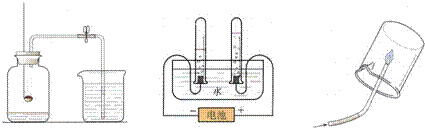
A���ⶨ�����е��������� B��ˮ���ʵ�� C���������ɷ�
A��B��C�����о�������ɵ�ʵ�飮���о�������֪��Aʵ�������ÿ�ȼ������ƿ�ڵ�
����
����
��ʹƿ��ѹǿ��С���Ʋ���������������ģ�Bʵ����ͨ��֤���������
Ԫ������
Ԫ������
���ƶ�ˮ����ɵģ���Cʵ����
B
B
����ʵ����ţ�ʵ��ķ�����ͬ��
��2��̽��2���о�������������--���������������Ƿ�պ���ȫ�к�
С���ò�����պȡ��Ӧ�����Һ����pH��ֽ�ϣ����ָ���Һ��pH��7��˵���������������������֤һ�£�
| ʵ������ |
ʵ������ |
��������� |
| ȡ��Ӧ��������Һ���Թ��У� ����п�� ����п�� |
�����ݲ��� �����ݲ��� |
֤���о�������ȷ |
��3��̽��3��ijͬѧ����֤�������ǵ���Ҫ�ɷ���̼���Σ����ռ����������塱��ʵ�飮��������·�������ʵ�飺
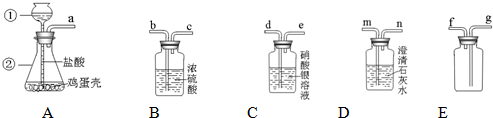
�ٸ���������ѧ��֪ʶ������ΪAװ�ú�
D
D
װ������������ʲô����ʱ������˵�������ǵ���Ҫ�ɷ���̼���Σ�
Dװ���г���ʯ��ˮ�����
Dװ���г���ʯ��ˮ�����
��д����װ���з�����Ӧ�Ļ�ѧ����ʽ��
CO2+Ca��OH��2=CaCO3��+H2O
CO2+Ca��OH��2=CaCO3��+H2O
��
�ڸ�ͬѧ��Ҫ�ռ�һƿ�����ĸ����壬��һ����֤�������ʣ���������װ�õ�˳���ǣ�����д���ӿ���ĸ��
a��d��e��b��c��g
a��d��e��b��c��g
�����У�Cװ�õ������ǣ�
��ȥCO2�л��е�HCl����
��ȥCO2�л��е�HCl����
��
��4��ij�о���ѧϰС����Э����ʦ����ʵ�鴢����ʱ������һ����Ŷ�����������ƣ�Ϊ������������������������̽����ȡ����������Ʒ11.4g����ƿ�У�����38.6gˮ�����γ�����Һ�����ڵ�����ƽ�ϣ�����ƿ����εμ�14.6%��ϡ���ᣬ���ȡ��������ͼ����ʾ����ʵ���ü���ϡ�������������ƿ�����ʵ�������ϵ��ͼ����ʾ��
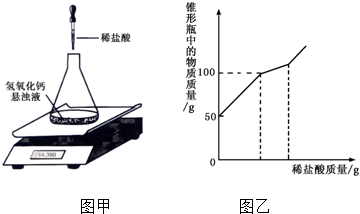
��11.4g����Ʒ�и��ɷֵ�������




 ����5��2���ϵ�д�
����5��2���ϵ�д�
 ��
��



