
 x=2.33g
x=2.33g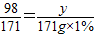 y=0.98g
y=0.98g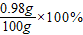 =0.98%
=0.98%


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011��ȫ���п���ѧ����������18 ������Դ�����úͱ��� ���ͣ��ʴ���
��2011���Ĵ��ϳ䣬17�⣩CO��һ����ɫ����ζ���ж��Ŀ�ȼ������Ⱦ���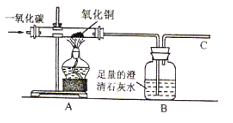
�л�ԭ�ԣ�������ұ��ҵ��ij��ȤС��Ϊ̽������л�ԭ�ԣ����������ʵ��װ�á����
�������й����⣺
��CO������ʹ���ж�����ԭ����
��ʵ�鿪ʼʱ����ͨ��COһ��ʱ����ټ��ȣ���ԭ����
��ʵ����ͼA���������з����������� ��
�仯ѧ��Ӧ����ʽΪ
��ʵ����ͼB��������������
�仯ѧ��Ӧ����ʽΪ
�ݸ�װ������һ�����Ե�ȱ�ݣ���ָ����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011��ȫ���п���ѧ����������21 ������������ ���ͣ�������
��2011���Ĵ��ϳ䣬18�⣩ij��ѧ��ѧ����С���ij�����ŷŵĺ���������ķ�ˮ���г�����⡣ȡһ�����ķ�ˮ������������Ϊ1%������������Һ�кͣ��������ʲ�������������Ӧ������ǡ���к�ʱ����������������Һ������Ϊ171g�����˺����Һ������Ϊ268.67g���������ǹ���ʱ��Һ������ʧ����������������λС����
�ٷ�Ӧ�����ɳ���������Ϊ���٣�
�ڸù����ŷŵķ�ˮ�������������������Ϊ���٣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011���Ĵ�ʡ�ϳ����п���ѧ�Ծ��������棩 ���ͣ������

�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com