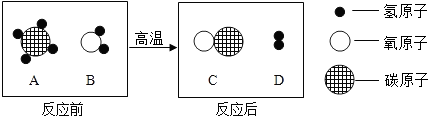
��2013?���ݣ���ʵġ�̼�������˵����������һ���߽���̼�������磮
��1�����ݱ�1�ṩ����Ϣ����д�йغ�̼���ʵĶ�Ӧ���ԣ�
| ������; |
���ʯ�и�� |
ʯī���缫 |
����̿��ˮ |
| ��Ӧ���� |
�� Ӳ�ȴ� Ӳ�ȴ� |
�� ������ ������ |
�� ������ ������ |
��2��Һ̬������̼������������˾ȵ��������ҷ����Ļ��֣�����˵����ȷ����
AB
AB
�����ţ���
A��Һ̬������̼��������Ⱦ��������
B��������̼�ɸ�����ȼ������棬��������
C��Һ̬������̼����ʱ���ȣ������˿�ȼ����Ż��
��3������Ķ�����̼�Ӿ��ˡ�����ЧӦ����д��һ�����ٶ�����̼�ŷŵĽ���
��Լ��ֽ
��Լ��ֽ
��
��4��������̼��һ�ֱ������Դ���̶������ö�����̼��һ���ɹ������ǣ��ڸ��¸�ѹ�£�CO
2��NH
3���Ժϳ�����[CO��NH
2��
2]��ͬʱ����ˮ���÷�Ӧ�Ļ�ѧ����ʽΪ
��
��5��Ŀǰ�������Ի�ʯȼ��Ϊ��Ҫ��Դ����ʯȼ����ú��
ʯ��
ʯ��
����Ȼ�������Ƕ�����
��������
��������
�������������������������Դ��
��6����Ȼ���м�����ȫȼ�յĻ�ѧ����ʽΪ
��
��7���ӱ����ݷ�������ú��ȣ�����Ȼ����ȼ�ϵ��ŵ���
����������Ȼ����ú��ȫȼ�գ���Ȼ��ȼ�ղ���������̼������С��ú���ų�����������ú
����������Ȼ����ú��ȫȼ�գ���Ȼ��ȼ�ղ���������̼������С��ú���ų�����������ú
��
|
1g������ȫȼ�� |
| ����������̼������/g |
�ų�������/kJ |
| ���� |
2.75 |
56 |
| ̼ |
3.67 |
32 |

 ��ʵġ�̼�������˵����������һ���߽���̼�������磮
��ʵġ�̼�������˵����������һ���߽���̼�������磮


 ��2013?���ݣ���ʵġ�̼�������˵����������һ���߽���̼�������磮
��2013?���ݣ���ʵġ�̼�������˵����������һ���߽���̼�������磮 ��ʵġ�̼�������˵����������һ���߽���̼�������磮
��ʵġ�̼�������˵����������һ���߽���̼�������磮