
ȥ�궬��ͽ��ҹ����������������Ϻܶ�ʡ�����ܰ��겻�����ش�ɺ���һЩ����Ⱥ����ˮ�������ѣ������������İ����£��Ѱ��ˮԴ��
(1)�����ǵ�ˮ������ ������ ����������ˮ��У���ˮ�г��ֽ϶�ˮ������ˮΪ (ѡ�Ӳˮ������ˮ��
������ ����������ˮ��У���ˮ�г��ֽ϶�ˮ������ˮΪ (ѡ�Ӳˮ������ˮ�� )��
)��
(2)�������мӴ�Ա�����ˮ�ʵ��������ȣ�����˵�����ˮ����Ⱦ��ԭ��֮һ��
��
(3)ȫ�������ů�ᵼ�¸ɺ������Ե��ֺ���Ƶ�������ȫ�������ů���Ͼ���ս��������Ӧ��ȡ�Ĵ�ʩ��  (дһ������)��
(дһ������)��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
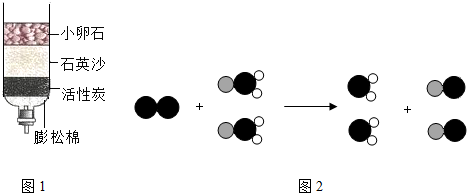
 ��ʾ��ԭ�ӣ�
��ʾ��ԭ�ӣ� ��ʾ��ԭ�ӣ�
��ʾ��ԭ�ӣ� ��ʾ��ԭ�ӣ�����д����Ӧ�Ļ�ѧ����ʽ
��ʾ��ԭ�ӣ�����д����Ӧ�Ļ�ѧ����ʽ�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
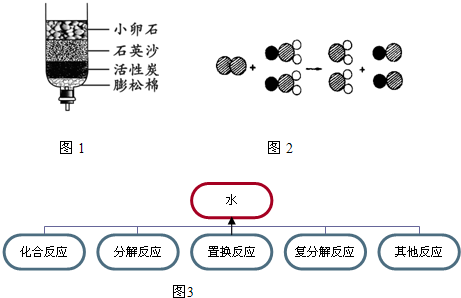
 ��ʾ��ԭ�ӣ�
��ʾ��ԭ�ӣ� ��ʾ��ԭ�ӣ�
��ʾ��ԭ�ӣ� ��ʾ��ԭ�ӣ�����д����Ӧ�Ļ�ѧ����ʽ
��ʾ��ԭ�ӣ�����д����Ӧ�Ļ�ѧ����ʽ
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com