
 (2)����ͼ��ˮͨ��ֽ��ʾ��ͼ����ʵ������У��Թ�a�в����������� ��д���÷�Ӧ�Ļ�ѧ����ʽ��
(2)����ͼ��ˮͨ��ֽ��ʾ��ͼ����ʵ������У��Թ�a�в����������� ��д���÷�Ӧ�Ļ�ѧ����ʽ�� ![]() ��
��
(3)Ϊ��ȥˮ�еIJ��������ʣ�ijͬѧ��������ͼ��ʾ�ļ���ˮ�������л���̿����Ҫ������ ��
(4)ijЩ�ط�������ˮ�к���������Ca(HCO3)2�ȿ������Ρ���ˮʱ��Ca(HCO3)2�����ֽⷴӦ�����������Ե�CaCO3����д��Ca(HCO3)2���ȷֽ�Ļ�ѧ����ʽ ��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2012�콭��ʡ��ͨ�о��꼶��ѧ����ĩ����ģ�⻯ѧ�Ծ� ���ͣ������
ˮ������֮Դ��Ҳ������������Դ��������ѧ���Ļ�ѧ֪ʶ���ش��������⣺
(1) ��ѧ���š�3H2O����������
(2)����ͼ��ˮͨ��ֽ��ʾ��ͼ����ʵ������У��Թ�a�в��������������� ��д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
(3) ��ȥˮ�������Թ������ʳ��õIJ����� ��Ϊ�˾���ˮ��ijͬѧ��������ͼ��ʾ�ļ���ˮ�������������˻���̿�� �ԡ�
��4������ˮ��Ӳˮ������ˮ�����õ������� ������˵��������ʹӲˮ������һ�ֳ��÷��� ��
��5�����곤�������ε��������˰��꺱���ĸɺ����ɿݵij���ʱʱɢ���Ŷ���������ŵ���ζ����Ҫԭ���� ��
��6��ʹ������ϴ�·������ڱ���ˮ��Դ������ġ��ס���ָ ����ѡ�ԭ�ӡ��������ӡ��������ӡ�����Ԫ�ء���
��7����дһ��ˮ������ ��
��8������Դ�Ƕȿ���ˮ������֮Դ���������ϲ���������ĸ����ǣ�
�����������дһ���ˮ����һ����ˮ��ʩ����
(9)ijЩ�ط�������ˮ�к���������Ca(HCO3)2�ȿ������Ρ���ˮʱ��Ca(HCO3)2�����ֽⷴӦ�����������Ե�̼��ơ�ˮ�Ͷ�����̼������д��Ca(HCO3)2���ȷֽ�Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�꽭��ʡ��ͨ�о��꼶��ѧ����ĩ����ģ�⻯ѧ�Ծ� ���ͣ������
ˮ������֮Դ��Ҳ������������Դ��������ѧ���Ļ�ѧ֪ʶ���ش��������⣺
(1) ��ѧ���š�3H2O����������
(2)����ͼ��ˮͨ��ֽ��ʾ��ͼ����ʵ������У��Թ�a�в��������������� ��д���÷�Ӧ�Ļ�ѧ����ʽ�� ��

 (3) ��ȥˮ�������Թ������ʳ��õIJ�����
��Ϊ�˾���ˮ��ijͬѧ��������ͼ��ʾ�ļ���ˮ�������������˻���̿�� �ԡ�
(3) ��ȥˮ�������Թ������ʳ��õIJ�����
��Ϊ�˾���ˮ��ijͬѧ��������ͼ��ʾ�ļ���ˮ�������������˻���̿�� �ԡ�
��4������ˮ��Ӳˮ������ˮ�����õ������� ������˵��������ʹӲˮ������һ�ֳ��÷��� ��
��5�����곤�������ε��������˰��꺱���ĸɺ����ɿݵij���ʱʱɢ���Ŷ���������ŵ���ζ����Ҫԭ���� ��
��6��ʹ������ϴ�·������ڱ���ˮ��Դ������ġ��ס���ָ ����ѡ�ԭ�ӡ��������ӡ��������ӡ�����Ԫ�ء���
��7����дһ��ˮ������ ��
��8������Դ�Ƕȿ���ˮ������֮Դ���������ϲ���������ĸ����ǣ�
�����������дһ���ˮ����һ����ˮ��ʩ����

(9)ijЩ�ط�������ˮ�к���������Ca(HCO3)2�ȿ������Ρ���ˮʱ��Ca(HCO3)2�����ֽⷴӦ�����������Ե�̼��ơ�ˮ�Ͷ�����̼������д��Ca(HCO3)2���ȷֽ�Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ˮ������֮Դ��Ҳ������������Դ��������ѧ���Ļ�ѧ֪ʶ
�ش��������⣺
(1)��Լˮ��Դ����ֹˮ��Ⱦ��ÿ������Ӧ�������κ������������������ˮ����Ⱦ���У�����ţ� ��
A����ҵ��ˮֱ���ŷ� B����ҵ�����������ŷ�
C����ֹʹ�ú���ϴ�·� D������ʹ�û��ʡ�ũҩ
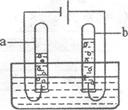
(2)����ͼ��ˮͨ��ֽ��ʾ��ͼ����ʵ������У��Թ�a�в����������� ��д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
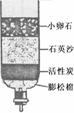
(3)Ϊ��ȥˮ�еIJ��������ʣ�ijͬѧ��������ͼ��ʾ�ļ���ˮ�������л���̿����Ҫ������ ��
(4)ijЩ�ط�������ˮ�к���������Ca(HCO3)2�ȿ������Ρ���ˮʱ��Ca(HCO3)2�����ֽⷴӦ�����������Ե�CaCO3����д��Ca(HCO3)2���ȷֽ�Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ˮ������֮Դ��Ҳ������������Դ��������ѧ���Ļ�ѧ֪ʶ���ش��������⣺
(1)��Լˮ��Դ����ֹˮ��Ⱦ��ÿ������Ӧ�������κ������������������ˮ����Ⱦ���У�����ţ� ��
A����ҵ��ˮֱ���ŷ� B����ҵ�����������ŷ�
C����ֹʹ�ú���ϴ�·� D������ʹ�û��ʡ�ũҩ
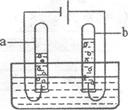
(2)����ͼ��ˮͨ��ֽ��ʾ��ͼ����ʵ������У��Թ�a�в����������� ��д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
(3)Ϊ��ȥˮ�еIJ��������ʣ�ijͬѧ��������ͼ��ʾ�ļ���ˮ�������л���̿����Ҫ������ ��
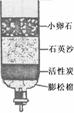 (4)ijЩ�ط�������ˮ�к���������Ca(HCO3)2�ȿ������Ρ���ˮʱ��Ca(HCO3)2�����ֽⷴӦ�����������Ե�CaCO3����д��Ca(HCO3)2���ȷֽ�Ļ�ѧ����ʽ ��
(4)ijЩ�ط�������ˮ�к���������Ca(HCO3)2�ȿ������Ρ���ˮʱ��Ca(HCO3)2�����ֽⷴӦ�����������Ե�CaCO3����д��Ca(HCO3)2���ȷֽ�Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com