
ʵ������һƿ�����Һ����ʦ��С��ͬѧ��Ʒ����ⶨ�÷�Һ�����������������С��ͬѧ��ȡһ����С�ձ�����������Ϊ18.2g��Ȼ�������е������������Һ�������������Ϊ33.2g��֮��һö����Ϊ10.8g������������ɰֽɰȥ���⣩�����С�ձ��з�Ӧ�����������治�������ݲ������ٴγ�����������Ϊ43.9g����ش��������⣺
��1����Ӧ�в���������������� ��
��2������÷�Һ�����������������д��������̣�����������һλС������
��3���������������δ�������ԣ�2����������Ӱ���� ��ѡ�ƫ����ƫС��������Ӱ�족����ԭ���� ��
��1��0.1g ��2��32.7%. ��3��ƫС������Ҳ������һ�������ᡣ
���������������1������ϡ���ᷴӦ����������������������ȫ��Ӧ�������е����ʵ������������������������������������Ϊ��33.2g+10.8g-43.9g=0.1g�����0.1g��
��2�� ��������Ӧ�����������Ϊx��
�ձ���ϡ�����Һ������Ϊ��33.2g-18.2g=15.0g
Fe+H2SO4�TFeSO4+H2��
98 2
x 0.1g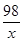 ��
��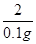
X=4.9g
�÷�Һ���������������Ϊ��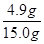 ��100%=32.7%
��100%=32.7%
�𣺷�Һ�����������������32.7%��
��3���������������δ����������������һ�������ᣬ�����������ᷴӦ������������ˮ����Ӧ����ʽΪFe2O3+3H2SO4=Fe2��SO4��3+3H2O�������������������������ƫС���Ӷ�ʹ��������������������ƫС�����ƫС������Ҳ������һ�������ᡣ
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ��㣻�й��������������ļ��㣻��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ


 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д� �����ܿ����ϵ�д�
�����ܿ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��9�֣�ij���᳧��ˮ�к����������ᣮ�ŷ�ǰ������ԱС���ȶԷ�ˮ������ĺ������м�⡣
��1��������Һ��
������������������Ϊ4%��NaOH��Һ100g����ҪNaOH���� g��ˮ ml
��ˮ���ܶ�Ϊ1 g/CI113����
��2����������
ȡ��ˮ��Ʒ98g����������μ���NaOH��Һ��ǡ����ȫ��Ӧʱ������4%��NaOH
��Һ20g�������ˮ�������ɷ־�����NaOH��Ӧ�����Լ����ˮ���������������
��д��������̣���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
С�մ�(��Ҫ�ɷ�ΪNaHCO3)�г����������Ȼ��ơ���ѧ��ȤС���ͬѧΪ�˲ⶨijƷ��С�մ���NaHCO3����������������������ʵ�飺������Ʒ�����ձ��У������������μ�ϡ���ᣬ�����ٲ�������Ϊֹ����õ��й��������±���ʾ��
| ���� | ��Ʒ | ���ĵ�ϡ���� | ��Ӧ�����Һ |
| ����(g) | 9 | 75.4 | 80 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��ij����������̼�ĺϽ���Ʒ5��8��Ͷ�뵽100��ϡ������ǡ����ȫ��Ӧ��ʣ���ɫ������0��2�ˣ����ϡ��������������Ƕ��٣�(���ԭ������H:12, Cl:35��5, Fe :56 )
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
ij��ѧ��ȤС��Ϊ�˴��Բⶨһ��ʯ��ʯ��Ʒ��CaCO3��������������Ʋ���������ʵ�顣
ʵ��װ����ͼ��ʾ����ȡ��ϸ��2.60gʯ��ʯ��Ʒ����4�μ���ϡ���ᣬ��ַ�Ӧ�����ٲ�������Ϊֹ����÷�Ӧǰ����й����������
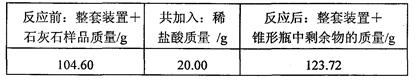
��1������ʯ��ʯ��Ʒ��CaCO3������������
��2����������ʵ�������ʯ��ʯ��Ʒ��������Һ��������������ͼ��װ���жϣ���ʵ�������� ���ƫ����ƫС����ȷ������ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��3.6g̼��Ƹ�������һ��ʱ�����ȴ����ù���ʣ�����и�Ԫ�ص���������Ϊ57.6%,����������������Ƶ�������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
ʵ���ҳ��ô���ʯ��ϡ���ᷴӦ����ȡ������̼������25g��̼���Ϊ80%�Ĵ���ʯ��200gϡ���ᷴӦ��ȡ������̼��ǡ����ȫ��Ӧ���Լ��㣺
��1�����ɶ�����̼�������Ƕ��٣���2������ϡ������������������Ƕ��٣�
��3����Ӧ��������Һ�����������Ƕ��٣�(��������ȷ��0.1%)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
����ʯ��ʯ���������ڲⶨʯ��ʯ��CaCO3 ��������ͼ��ij��ʯ��ʯ���չ����������ı仯����ͼ���������������Ȳ��ֽ⣩����д��������̣�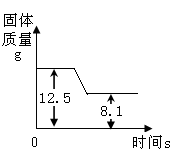
��1��д��ʯ��ʯ�ֽ�Ļ�ѧ����ʽ ��
��2����ʯ��ʯ��CaCO3�����ʵ����� mol��
��3����ʯ��ʯ��CaCO3 ���������� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com