
 3B+5CO2+19H2O
3B+5CO2+19H2O ×100%�����з������
×100%�����з������ 3B+5CO2+19H2O����Ӧ����̼���⡢������ԭ�Ӹ����ֱ�Ϊ5��38��29��6����Ӧ�����������̼���⡢������ԭ�Ӹ����ֱ�Ϊ5��38��29��0�����ݷ�Ӧǰ��ԭ�����ࡢ��Ŀ���䣬��3B�к���6����ԭ�ӣ���ÿ��B������2����ԭ�ӹ��ɣ�������X�Ļ�ѧʽΪN2��
3B+5CO2+19H2O����Ӧ����̼���⡢������ԭ�Ӹ����ֱ�Ϊ5��38��29��6����Ӧ�����������̼���⡢������ԭ�Ӹ����ֱ�Ϊ5��38��29��0�����ݷ�Ӧǰ��ԭ�����ࡢ��Ŀ���䣬��3B�к���6����ԭ�ӣ���ÿ��B������2����ԭ�ӹ��ɣ�������X�Ļ�ѧʽΪN2�� ×100%=50%��
×100%=50%��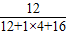 =96g��
=96g��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| 200kPa |
| ||
| 200kPa |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ��κ�������Ч����Լ��ʹ��ú��ʯ�͡���Ȼ����������������Ҫ�Ļ�ʯȼ�ϣ��ӽ��������������������Դ��ȫ�������ٵĹ�ͬ���⣬�Ƿ�չ��̼���õĵ���֮�����ش��������⣺
��κ�������Ч����Լ��ʹ��ú��ʯ�͡���Ȼ����������������Ҫ�Ļ�ʯȼ�ϣ��ӽ��������������������Դ��ȫ�������ٵĹ�ͬ���⣬�Ƿ�չ��̼���õĵ���֮�����ش��������⣺
| ||
| ���� |
| �ϳ��� |
| CO��H2 |
| ||
| ||
| ||
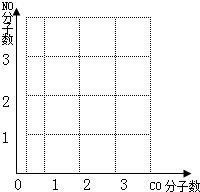
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ||
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com