
ij���Ƽ�����Ҫ�ɷ���̼��ơ�Ϊ�ⶨ�ò��Ƽ���̼��Ƶ�����������ijͬѧ��ȡһ����������Ʒ�����ձ�������м���ϡ���������ٲ�������Ϊֹ�����ˣ��õ�����2��5g��ͬʱ�õ���Һ100�ˣ����ⶨ����Һ��������������Ϊ 11��1%�����㣺
��1���ò��Ƽ���̼��Ƶ�����������
��2��ij�˳�������ʳ֮�⣬ÿ���貹��0��64g��Ԫ�أ���ÿ����Ҫ�������ֲ��Ƽ����ٿˣ�
��3���������õ�����Һ����������������Сһ�룬Ӧ��ˮ���ٿˣ�
��1��80% ��2�� 2g ��3��100g
��������������⣺��1����ò��Ƽ���̼��Ƶ�����ΪX
2HCl+ CaCO3 = CaCl2 +H2O+CO2
100 111
x 100gX11��1%
100��x = 111��100gX11��1% ?x=10g
CaCO3 %=10/(10+2��5) X100%=80%
����0��64g��Ԫ�أ���ÿ����Ҫ�������ֲ��Ƽ�������Ϊ��0��64/80%/40%=2g
�裺�����õ�����Һ����������������Сһ�룬Ӧ��ˮy��
100 *11��1% =(y+100)*11��1%/2
y=100g
���㣺�йػ�ѧʽ����ѧ����ʽ����Һ�������������ļ���


 53���ò�ϵ�д�
53���ò�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��ʵ����������������������Ϊ10%���Ȼ�����Һ100g�����ȡ�Ȼ������� �ˡ�
�Ƴ�ȡ�Ȼ�����̼���ƵĹ�������22.3�˷����ձ��У������Ȼ�����Һ��ǡ����ȫ��Ӧ����Ӧ�Ļ�ѧ����ʽΪBaCl2 + Na2CO3 = BaCO3�� + 2NaCl��������Ӧ��ȫ����ˣ��ð�ɫ����19.7�˺���Һ200g���ٹ�������������̼���Ƶ�������
����Һ���������ʵ���������?
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��ľ����һ��ũ�ҷʣ�������Ҫ�ɷ���̼��أ���������أ��Ȼ��صȡ���ѧ��ȤС��Ϊ�ⶨij��ľ����Ʒ�е���Ч�ɷ֣�ȡ50g��ľ�����ձ��У����ϵ���������Һ��������30g������Һʱ�����������ݲ�������ʱ�ձ��еIJ������������Ϊ77.8g���������ľ�ҵ������ɷֲ�����Ԫ���Ҳ����ᷴӦ��
�����ش�
��1����ľ�ҵ���Ҫ�ɷ��������������Ϊ____________����ᡢ����Ρ�����
��2����ͼ��ʾ��Ӧ�����зų����������������������Һ�Ĺ�ϵ���ߣ������ͼ����������a����ֵ��a=________________g��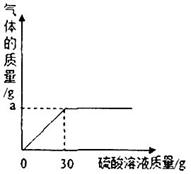
��3�������ľ����Ʒ��̼��ص�������������Ҫ��д��������̣�
��4��ͨ������ʵ�飬��ø�50g��ľ���л��������������Ϊ8.7g���Ȼ��ص�����Ϊ1.49g�����50g��ľ����Ʒ�м�Ԫ�ص�����Ϊ__________g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��4�֣���ѧʵ���ᳫ��ɫ��������ʵ��װ�ý����ͻ��Ľ���һ���ܺõ�;����ͼ����ʵ������ȡ������CO2��װ�ã�ͼ���Ƕ�ͼ��ʵ��װ�õġ��͡����Ľ����װ�á� 
��1��ͼ���������ϵι���ʵ���е�������ͼ���е� ������ͬ������ĸ��ţ���
��2��ͨ���ü�װ����ɸ�ʵ����Ҫ�������ǡ��͡�ʵ��װ��������10�������á��͡�ʵ��װ�þ��е��ŵ��� ��
��3������װ����ɡ���ȡ������CO2����ʵ�飬������0.73g10%�����ᡣ�����ʵ������в���CO2�������Ƕ��ٿˣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
(3��)��Na2CO3��NaCl��������32.9g�����ձ��У���ʱ������Ϊ202.9g������326.9g���ᣬǡ����ȫ��Ӧ����û�������ݳ����ٴγ�����������Ϊ521.0g������������Һ�����ʵ���������(CO2���ܽ���Բ���)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��3�֣���ҵ��ʹ�á�������Cr������������߸�����Ʒ�Ŀ���ʴ����������һ����Ҫ��Ӧ�Ļ�ѧ����ʽ��Fe + CrCl2 =" Cr" + FeCl2����֪�μӷ�Ӧ��������Ϊ112 kg���������ɸ���������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��3�֣���ij������Ʒ�У�����������������Ϊ10%������ͭ��Һ160 g��ǡ����ȫ��Ӧ����Ʒ�����ʲ�����ˮҲ��������ͭ��Ӧ��������ͼ�ṩ�����ݼ��㡣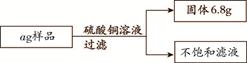
��1��a����ֵ��
��2��������Һ�м���40.8 gˮ����������Һ�����ʵ���������.
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
ijУ��ѧʵ����Ҫ�ⶨ����������Һ�������������������ʵ������ͼ��ʾ��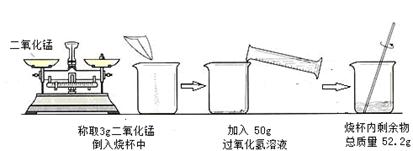
����㣺
��1������������������
��2������������Һ����������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
Ϊ�ⶨij������Ʒ��������ľ̿�ۣ������ĺ�������ʢ��219gϡ������ձ��в��ϼ���������Ʒ�����������������������������Ʒ�����Ĺ�ϵ��ͼ����ʾ��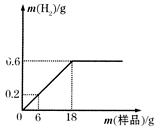
��1��ϡ����ǡ����ȫ��Ӧʱ����������������Ϊ g��
��2��6g������Ʒ�к������ʵ�����Ϊ g��
��3����ϡ���������ʵ�����������д��������̣���
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com