
��7�֣�ijʵ��С�齫����Ƥ����ԭ����ʯ�ҡ������ʳ�η�������ˮ�У���ַ�Ӧ����ˣ��õ�������Һ��������
��1�����������з�����Ӧ�Ļ�ѧ����ʽΪ ��
��2���ҵ��Ʋ��ǣ�����Һ��һ�����ڵ������� ��
��3��Ϊ��һ��ȷ������Һ���п��ܵ���ɣ�ͬѧ���������ͼ��ʾ��ʵ��װ�ý�����֤��
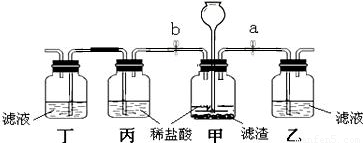
�ټ�װ���з�����Ӧ�Ļ�ѧ����ʽΪ ��
�ڹر�ֹˮ��b����ֹˮ��a�����۲쵽��װ�ó��ֻ�����������Һ�е������� ��
�۹ر�ֹˮ��a����ֹˮ��b��ʵ��Ŀ���� �������� ��ʵ�����ɵó��д����ʵĽ��ۡ�
������Һ��ֻ�����Ʋ��е����ʣ�ͬʱ��a��bʱ������������ͽ����� ��
��1��CaO + H2O Ca(OH)2 Ca(OH)2 + Na2CO3 CaCO3��+ 2NaOH
��2��NaCl��NaOH ��3�� ��CaCO3 + 2HCl CaCl2 + H2O + CO2�� ��NaCl ��NaOH ��Ca(OH)2
����֤������Һ���Ƿ���̼���� ����Һ�������������ݲ���
�ܶ��������ݲ��������в����ǣ�������Һ��ֻ��NaCl��NaOH��
����������1������������ˮ��Ӧ�����������ƣ�������������̼���Ʒ�Ӧ����̼��ƺ��������ƣ����Ա����Ϊ��CaO+H2O�TCa��OH��2��Ca��OH��2+Na2CO3�TCaCO3��+2NaOH��
��2�����ݷ�Ӧ��֪���������������ƣ��Ȼ��Ʋ����뷴Ӧ������������Һ��һ�����������ƺ��Ȼ��ƣ����Ա����Ϊ��NaCl��NaOH��
��3����̼����������ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼�����Ա����Ϊ��CaCO3+2HCl�TCaCl2+H2O+CO2����
�ڹر�ֹˮ��b����ֹˮ��a��������Ķ�����̼�������У���װ�ó��ֻ�������˵����Һ�к����������ƣ����Ա����Ϊ��NaCl��NaOH��Ca��OH��2��
�۹ر�ֹˮ��a����ֹˮ��b�������Ķ�����̼�ܰ�����ѹ�붡�У���������̼���Ʒ�Ӧ��������Ӷ�����̼���ƵĴ��ڣ����Ա����Ϊ����֤������Һ�ﺬ��̼���ƣ�����Һ�������������ݲ�����
��ͬʱ��a��b���������ܽ����ң��ܽ�����ѹ�붡�����Ҳ�������Ҷ������ݣ���֤��ֻ�����Ȼ��ƺ��������ƣ����Ա����Ϊ�����������ݲ��������в����ǣ�������Һ��ֻ��NaCl��NaOH��


 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д� �������ϵ�д�
�������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ʵ����� ���������Լ��Ļ�ѧʽ�� | ʵ������ | ʵ����� | ����Һ����� |
 | |||
 | |||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ʵ����� ���������Լ��Ļ�ѧʽ�� |
ʵ������ | ʵ����� | ����Һ����� |
 |
���������� |
��Na2CO3 |
NaOH��NaCl��Na2CO3 |
 |
�а�ɫ�������� |
��Ca��OH��2 |
NaOH��NaCl��Ca��OH��2 |
�ۺ���������ʵ�� |
������ʵ������������� |
����Na2CO3Ҳ��Ca��OH��2 |
NaOH��NaCl |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ʵ����� ���������Լ��Ļ�ѧʽ�� |
ʵ������ | ʵ����� | ����Һ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ʵ����� ���������Լ��Ļ�ѧʽ�� |
ʵ������ | ʵ����� | ����Һ����� |
 HCl HCl |
���������� |
��Na2CO3 |
Na2CO3��NaCl��NaOH Na2CO3��NaCl��NaOH |
 Na2CO3 Na2CO3 |
������� |
��Ca��OH��2 |
Ca��OH��2��NaCl��NaOH Ca��OH��2��NaCl��NaOH |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com