Ęū³µŹĒĻÖ“śÉś»īÖŠ²»æÉȱɣµÄ“ś²½¹¤¾ß£®Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©Ęū³µµēĀ·ÖŠµÄµ¼Ļ߶ąŹżŹĒÓĆĶÖĘ×öµÄ£¬ÕāŹĒĄūÓĆĮĖ½šŹōĶµÄµ¼µēŠŌŗĶ
ŃÓÕ¹
ŃÓÕ¹
ŠŌ£®
£Ø2£©ĻĀĮŠĘū³µÅ伞¼°ÓĆĘ·ÖŠ£¬ÓĆŗĻ½šÖĘ×öµÄŹĒ£ØĢī×ÖÄø£©
a
a
£®a£®øÖĢśĶāæĒb£®²£Į§³µ“°c£®Ļš½ŗĀÖĢ„d£®Õęʤ׳ŅĪ
£Ø3£©ĢśŌŚ³±ŹŖµÄæÕĘųÖŠČŻŅ׊āŹ“£®
¢ŁĘū³µ±ķĆęÅēĘį£¬æÉŅŌŃÓ»ŗĘū³µµÄŠāŹ“£¬Ęä·ĄŠāŌĄķŹĒøō¾ųæÕĘų£ØŃõĘų£©ŗĶ
Ė®
Ė®
£®
¢ŚÅēĘįĒ°Šč½«ĢśÖĘĘ··ÅČėĻ”ŃĪĖįÖŠ³żŠā£ØĢśŠāÖ÷ŅŖ³É·ÖŹĒFe
2O
3£©£¬·“Ó¦³õĘŚæɹŪ²ģµ½ČÜŅŗÓÉĪŽÉ«±äĪŖ»ĘÉ«£¬ÕāŹĒŅņĪŖÉś³ÉĮĖ
FeCl3
FeCl3
£ØĢī»ÆѧŹ½£©£»·“Ó¦½ųŠŠŅ»»į¶ł£¬ÓÖæÉŅŌ¹Ū²ģµ½ÓŠĪŽÉ«ĘųÅŻ²śÉś£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ
Fe+2HClØTFeCl2+H2”ü
Fe+2HClØTFeCl2+H2”ü
£®
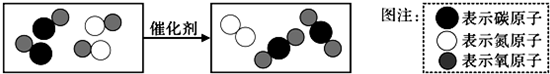
£Ø4£©”°Ī²Ęų“ß»Æ×Ŗ»ÆĘ÷”±æɽ«Ęū³µĪ²ĘųÖŠµÄÓŠ¶¾ĘųĢå×Ŗ»ÆĪŖĪŽ¶¾ĘųĢ壬Ķ¼±ķŹ¾ÖĪĄķĘū³µĪ²ĘųĖłÉę¼°·“Ó¦µÄĪ¢¹Ū¹ż³Ģ£® Ķ¼ÖŠĖłŹ¾·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ
£»




 æŚĖćĢāæر±¾©ø¾Å®¶łĶƳö°ęÉēĻµĮŠ“š°ø
æŚĖćĢāæر±¾©ø¾Å®¶łĶƳö°ęÉēĻµĮŠ“š°ø



