
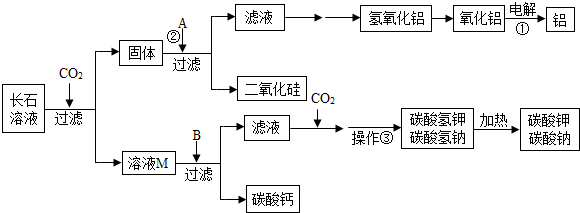
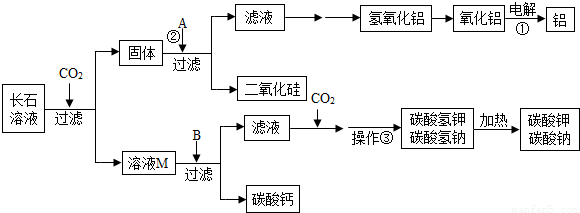
��������������•Ŭ�����Ƿ�����һ���кʹ���������̼���·������ǽ�������̼�ӻ������糧�������з�����������뺬�г�ʯ�ɷֵ�ˮ��Һ������кʹ������ڷ�Ӧ�����У���ʯ�ijɷֲ��������ն�����̼��ͬʱ��������һЩ���õĸ���Ʒ����Щ����Ʒ��������������ԭ���ϺͲ�������ҵ������Ҫ���̿�ʾ����ͼ��

��1�������ҽ�CO2�ӻ������糧�������з��������ͨ�뺬�г�ʯ��ˮ��Һ�����գ��ӻ����ĽǶȿ������������������� ����
��2����ʯ�ǵؿ�������Ŀ�ʯ�������ߴ�60%����ʯ��Ҫ�����س�ʯ��KAlSi3O8�����Ƴ�ʯ��NaAlSi3O8�����Ƴ�ʯ��CaAl2Si2O8����l t���Ƴ�ʯ�γɵ�ˮ��Һ������320kg CO2�����Ƴ�ʯ����CO2��Ӧ����ʽΪ
CaAl2Si2O8+2CO2+4H2O��Ca��HCO3��2+2SiO2��+2A1��OH��3������ģ�¸Ƴ�ʯ��д���Ƴ�ʯ����CO2�Ļ�ѧ��Ӧ����ʽ�� ����
��3��д����Ӧ�ٵĻ�ѧ����ʽ�� ������A��pHֵС��7����д��һ�����Ϸ�Ӧ�ڵĻ�ѧ����ʽ�� ����
��4����ҺM�е�����Ϊ�� ����B����������������������BΪ�� ������������ҪĿ��Ϊ�� ����
��1�����ٶ�����̼�ŷţ���������ЧӦ
��2��NaAlSi3O8+CO2+2H2O��NaHCO3+3SiO2��+Al��OH��3��
��3��2Al2O3 4Al+3O2��
Al��OH��3+3 HCl��AlCl3+3H2O
4Al+3O2��
Al��OH��3+3 HCl��AlCl3+3H2O
��4��Ca ��HCO3��2 NaHCO3 KHCO3����ʯ�ң��� Ca��OH��2��������Һ����ȡ����
��������
�����������1�������ж�����̼����������������ЧӦ�������ҽ�CO2�ӻ������糧�������з��������ͨ�뺬�г�ʯ��ˮ��Һ�����գ��ɼ��ٶ�����̼���ŷţ���������ЧӦ��
��2�����ݸƳ�ʯ���ն�����̼�Ļ�ѧ����ʽ�������еĸ�Ԫ�ػ�����Ԫ�أ���ȷд����Ӧ��ѧʽ��Ȼ����ƽ��ѧ����ʽ����NaAlSi3O8+CO2+2H2O��NaHCO3+3SiO2��+Al��OH��3����
��3����Ӧ����������ͨ�������������������������ѧ����ʽΪ2Al2O3 4Al+3O2����A��pHֵС��7���ܺ�������������Ӧ����A�������ᣬ���Ϸ�Ӧ�ڵĻ�ѧ����ʽ����Ϊ
4Al+3O2����A��pHֵС��7���ܺ�������������Ӧ����A�������ᣬ���Ϸ�Ӧ�ڵĻ�ѧ����ʽ����Ϊ
Al��OH��3+3HCl��AlCl3+3H2O��
��4����ʯ�к��мس�ʯ��KAlSi3O8�����Ƴ�ʯ��NaAlSi3O8�����Ƴ�ʯ��CaAl2Si2O8���Ƴ�ʯ��Һ��ͨ�������̼������̼����ء�̼�����ơ�̼����ƣ����˺�õ�����ҺM�к��е�����ΪKHCO3��NaHCO3��Ca(HCO3) 2���������ƿ�����������������������������KHCO3��NaHCO3��Ca(HCO3) 2��Ӧ����̼��Ƴ��������˺���Һ�к���̼���ơ�̼��أ�ͨ�������̼������̼�����ơ�̼����أ�Ҫ�õ�̼�����ƺ�̼����ع��壬�������ܼ�������Һ����ȡ�����־��壬Ȼ����Ⱥ�õ�̼���ơ�̼��ء�
���㣺�������ʵ��ת�����Ʊ�


 ����С��ʿ���������ϵ�д�
����С��ʿ���������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�꽭��ʡ�Ͼ����껨̨���п���ѧһģ�Ծ��������棩 ���ͣ������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com