
某化学兴趣小组同学用Na2CO3溶液和浓HCl来研究简易灭火器的反应原理时,对废液的成分进行探究。
【推理假设】 废液的成分中一定有 ,可能有Na2CO3或盐酸.
【实验探究】 确定废液成分中是否含有Na2CO3或盐酸
请你协助兴趣小组的同学完成实验探究,并完成实验报告。
| 实验步骤 | 现象 | 结论 |
| 该废液中一定没有盐酸 | ||
| 该废液中一定含有Na2CO3 |
 天天向上一本好卷系列答案
天天向上一本好卷系列答案 小学生10分钟应用题系列答案
小学生10分钟应用题系列答案科目:初中化学 来源: 题型:
兴趣小组同学向盛有生石灰的烧杯中加入碳酸钠溶液,搅拌、过滤、洗涤、干燥后得到固体A和溶液B,并对固体A和溶液B进行探究。
上述实验中发生的反应有:①CaO+H2O=Ca(OH)2;② (写化学方程式)。
(Ⅰ).固体A成分的探究及测定
[猜想]①固体A中只有CaCO3;②固体A中含有C aCO3和Ca(OH)2;
aCO3和Ca(OH)2;
含有Ca(OH)2的理由是 。
[实验] 取少量固体A于试管中加足量水,振荡、静置,向上层清液中滴加 硫酸铜溶液,出现 ,猜想②成立。
硫酸铜溶液,出现 ,猜想②成立。
[组成测定] 取8.7g固体A于试管中,加足量稀盐酸至固体完全溶解,收集到纯净CO2 2.2g,则固体A中CaCO3和Ca(OH)2的质量比为 。
(Ⅱ).溶液B的成分探究
[猜想]①溶液B中的溶质是NaOH;
②溶液B中的溶质是NaOH和Na2CO3;
③溶液B中的溶质是NaOH和Ca(OH)2。
根据对固体A成分的探究,小组同学认为猜想③正确,其理由是 。
[实验] 取少量溶液B于试管中,滴加 溶液,产生白色沉淀,证明猜想③正确。
查看答案和解析>>
科目:初中化学 来源: 题型:
下图是镁、锌、铁、铜四种金属与稀盐酸反应的示意图。
 |
(1)根据图中现象分析,A表示的金属是 。
(2)写出金属C与稀盐酸反应的化学方程式 。
(3)由图中现象可知,影响金属与酸反应快慢的因素有 。
查看答案和解析>>
科目:初中化学 来源: 题型:
下列有关溶液的说法错误的是( )
A. 溶液一定是无色透明的
B. 溶液由溶质和溶剂组成
C. 溶质可以是固体、液体和气体
D. 稀溶液也可能是饱和溶液
查看答案和解析>>
科目:初中化学 来源: 题型:
如图是元素周期表的一部分,请按要求填空.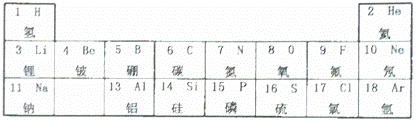
(1)由表中空格处元素组成的单质在空气中燃烧的化学方程式是 .
(2)表中不同元素最本质的区别是 (填字母).
A.各元素原子的质子数不同 B.各元素原子的中子数不同 C.各元素相对原子质量不同
(3)钠元素原子的结构示意图为,该原子在化学反应中易 (填“得到”或“失去”)电子,元素的化学性质主要由其原子的 决定.
(4)化学反应总是伴随着能量变化,锂是制作手机电池的原料,手机通话时电池中贮存的化学能转化为 .
查看答案和解析>>
科目:初中化学 来源: 题型:
下列物质的说法正确的是( )
A.鉴别纯棉织物和纯毛织物——各取少量纤维灼烧,闻气味
B.草木灰(K2CO3)是一种农家肥料,溶液显碱性 ,为增强肥效将其与氯化铵混合
C.吹灭燃烧着的蜡烛其灭火原理是隔绝氧气
D.用汽油除去衣服上的油污属于乳化作用
查看答案和解析>>
湖北省互联网违法和不良信息举报平台 | 网上有害信息举报专区 | 电信诈骗举报专区 | 涉历史虚无主义有害信息举报专区 | 涉企侵权举报专区
违法和不良信息举报电话:027-86699610 举报邮箱:58377363@163.com