
| Ũ���ᣨ�������� ��ѧʽ��H2SO4���� ��Է���������98 �ܶȣ�1.84g/cm3 ����������98% |
| ������\������ | OH- | NO3- | Cl- | SO42- | CO32- |
| H+ | �ܡ��� | �ܡ��� | �� | �ܡ��� | |
| Na+ | �� | �� | �� | �� | �� |
| Ba2+ | �� | �� | �� | ���� | ���� |
| ʵ��Ŀ�� | ʵ����� | ���� | ���ۻ�ѧ����ʽ |
��ȥ̼���� | ȡ�����ù�����Ʒ����ˮ�����Һ���μ�������______��Һ�� ��ַ�Ӧ����� | �а�ɫ�������� | �йط�Ӧ�Ļ�ѧ����ʽΪ ______ |
| �����Ƿ����������� | ����Һ�еμӷ�̪��Һ | ______ | ����Ʒ�к����������� |
 CaO+CO2����
CaO+CO2����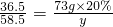 y=23.4g
y=23.4g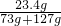 ��100%=11.7%
��100%=11.7% CaO+CO2����CaO+H2O�TCa��OH��2��
CaO+CO2����CaO+H2O�TCa��OH��2��| ʵ����� | ���� | ���ۻ�ѧ����ʽ |
| �Ȼ����������ᱵ�� | Na2CO3+BaCl2�TBaCO3��+2NaCl ����Na2CO3+Ba��NO3��2�TBaCO3��+2NaNO3�� | |
| ��̪��Һ���ɫ |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 18���ᡢ������й㷺��;����Ҫ�����
18���ᡢ������й㷺��;����Ҫ������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 �ᡢ������й㷺��;����Ҫ�����ij��ѧ�С���ͬѧΧ���⼸����������һϵ�е�̽�����
�ᡢ������й㷺��;����Ҫ�����ij��ѧ�С���ͬѧΧ���⼸����������һϵ�е�̽������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| Ũ���ᣨ�������� ��ѧʽ��H2SO4 ��Է���������98 �ܶȣ�1.84g/cm3 ����������98% |
| ������\������ | OH- | NO3- | Cl- | SO42- | CO32- |
| H+ | �ܡ��� | �ܡ��� | �� | �ܡ��� | |
| Na+ | �� | �� | �� | �� | �� |
| Ba2+ | �� | �� | �� | ���� | ���� |
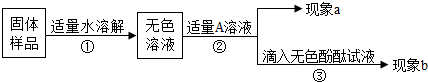
| ʵ��Ŀ�� | ʵ����� | ���� | ���ۻ�ѧ����ʽ |
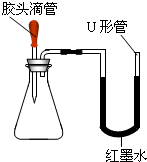
��ȥ̼���� |
ȡ�����ù�����Ʒ����ˮ�����Һ���μ������� ��ַ�Ӧ����� |
�а�ɫ�������� |
�йط�Ӧ�Ļ�ѧ����ʽΪ |
| �����Ƿ����������� | ����Һ�еμӷ�̪��Һ | ����Ʒ�к����������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011�����ʡ�������п���ѧһģ�Ծ��������棩 ���ͣ������

�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com