¼ŅÖŠÕōĀųĶ·ÓƵēæ¼īÖŠŗ¬ÓŠÉŁĮæNaCl£¬Ä³ŹµŃ銔×éŅŖ²ā¶ØøĆ“æ¼īÖŠNa2CO3µÄÖŹĮæ·ÖŹż£®Č”6g“æ¼īѳʷ·ÅČėÉÕ±ÖŠ£¬ÖšµĪ¼ÓČėĻ”ŃĪĖįÖĮ²»ŌŁ²śÉśĘųÅŻ£¬“ĖŹ±ÉÕ±ÖŠĆ»ÓŠ²»ČÜĪļ£¬¹²ĻūŗÄĻ”ŃĪĖį61.7g£¬²āµĆ·“Ó¦ŗóČÜŅŗµÄÖŹĮæĪŖ65.5g£®Ēė°ļÖśŹµŃ銔×éĶź³ÉŅŌĻĀ¼ĘĖć£ŗ
£Ø1£©Éś³É¶žŃõ»ÆĢ¼µÄÖŹĮæŹĒ¶ąÉŁ£æ
£Ø2£©ŃłĘ·ÖŠĢ¼ĖįÄʵÄÖŹĮæ·ÖŹżŹĒ¶ąÉŁ£Ø½į¹ū±£ĮōÖĮ0.1%£©£æ
£Ø3£©·“Ó¦ŗóČÜŅŗÖŠČÜÖŹµÄÖŹĮæ·ÖŹżŹĒ¶ąÉŁ£æ
½ā£Ø1£©øł¾ŻÖŹĮæŹŲŗć¶ØĀÉÉś³É¶žŃõ»ÆĢ¼µÄÖŹĮæĪŖ£ŗ6g+61.7g-65.5g=2.2g£»
£Ø2£©Éč£ŗѳʷ֊ŗ¬Ģ¼ĖįÄʵÄÖŹĮæĪŖx£¬·“Ӧɜ³ÉĀČ»ÆÄʵÄÖŹĮæĪŖy£®
Na
2CO
3+2HCL=2NaCl+H
2O+CO
2ӟ
106 117 44
x y 2.2g

x=
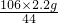
=5.3g

y=
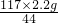
=5.85g
øĆѳʷ֊Ģ¼ĖįÄʵÄÖŹĮæ·ÖŹżĪŖ
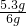
”Į100%=88.3%
£Ø3£©·“Ó¦ŗóČÜŅŗÖŠµÄČÜÖŹµÄÖŹĮæ·ÖŹżĪŖ
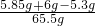
”Į100%=10%
“š£ŗÉś³É¶žŃõ»ÆĢ¼µÄÖŹĮæĪŖ2.2g£®ŃłĘ·ÖŠĢ¼ĖįÄʵÄÖŹĮæ·ÖŹżĪŖ88.3%£¬·“Ó¦ŗóČÜŅŗÖŠµÄČÜÖŹµÄÖŹĮæ·ÖŹżĪŖ10%£®
·ÖĪö£ŗ“æ¼īÖŠµÄĀČ»ÆÄĘÓėŃĪĖį²»·“Ó¦£¬¶ų“æ¼īÓėŃĪĖįÉś³É¶žŃõ»ÆĢ¼ĘųĢ壬ĖłŅŌĖłµĆČÜŅŗÖŹĮæ¼õÉŁĮĖ£¬Ėł¼õÉŁµÄ²æ·ÖĪŖ¶žŃõ»ÆĢ¼µÄÖŹĮ森ĄūÓƶžŃõ»ÆĢ¼µÄÖŹĮææÉĒóµĆĢ¼ĖįÄʵÄÖŹĮæŗĶ·“Ó¦ŗóČÜŅŗÖŠČÜÖŹµÄÖŹĮæ·ÖŹż£®
µćĘĄ£ŗŌŚ¼ĘĖć·“Ó¦ŗóČÜŅŗÖŠČÜÖŹµÄÖŹĮæ·ÖŹżŹ±£¬Ń§ÉśĶłĶłÖ»½«Ģ¼ĖįÄĘÓėŃĪĖį·“Ӧɜ³ÉµÄĀČ»ÆÄĘ¼ĘĖćŌŚÄŚ£¬¶ųŌĄ“¾ĶÓŠµÄĀČ»ÆÄĘĶü¼ĒĮĖ£¬ĖłŅŌŌŚ·ÖĪöŹ±Ó¦×¢ŅāŌĄ“¾ĶÓŠµÄĀČ»ÆÄʱŲŠė¼ÓČė£®
æĘÄæ£ŗ³õÖŠ»Æѧ
Ą“Ō“£ŗ2010-2011ѧğ°²»ÕŹ”°ö²ŗŹŠ»³Ō¶ĻŲ¾ÅÄź¼¶£ØÉĻ£©ĘŚÄ©»ÆѧŹŌ¾ķ£Ø½āĪö°ę£©
ĢāŠĶ£ŗ½ā“šĢā
¼ŅÖŠÕōĀųĶ·ÓƵēæ¼īÖŠŗ¬ÓŠÉŁĮæNaCl£¬Ä³ŹµŃ銔×éŅŖ²ā¶ØøĆ“æ¼īÖŠNa2CO3µÄÖŹĮæ·ÖŹż£®Č”6g“æ¼īѳʷ·ÅČėÉÕ±ÖŠ£¬ÖšµĪ¼ÓČėĻ”ŃĪĖįÖĮ²»ŌŁ²śÉśĘųÅŻ£¬“ĖŹ±ÉÕ±ÖŠĆ»ÓŠ²»ČÜĪļ£¬¹²ĻūŗÄĻ”ŃĪĖį61.7g£¬²āµĆ·“Ó¦ŗóČÜŅŗµÄÖŹĮæĪŖ65.5g£®Ēė°ļÖśŹµŃ銔×éĶź³ÉŅŌĻĀ¼ĘĖć£ŗ
£Ø1£©Éś³É¶žŃõ»ÆĢ¼µÄÖŹĮæŹĒ¶ąÉŁ£æ
£Ø2£©ŃłĘ·ÖŠĢ¼ĖįÄʵÄÖŹĮæ·ÖŹżŹĒ¶ąÉŁ£Ø½į¹ū±£ĮōÖĮ0.1%£©£æ
£Ø3£©·“Ó¦ŗóČÜŅŗÖŠČÜÖŹµÄÖŹĮæ·ÖŹżŹĒ¶ąÉŁ£æ
²éæ““š°øŗĶ½āĪö>>


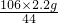 =5.3g
=5.3g
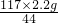 =5.85g
=5.85g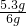 ”Į100%=88.3%
”Į100%=88.3%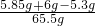 ”Į100%=10%
”Į100%=10%

 ѧĮ·æģ³µµĄæŚĖćŠÄĖćĖŁĖćĢģĢģĮ·ĻµĮŠ“š°ø
ѧĮ·æģ³µµĄæŚĖćŠÄĖćĖŁĖćĢģĢģĮ·ĻµĮŠ“š°ø