
| A�������� | B����չ�� | C�������� | D���������� |
 ��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�
��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����й㷺Ӧ�ø��ֽ�����
��1��Ŀǰ������һ�����ϵ�ͭ���ڵ����͵��Ź�ҵ����Ҫ����Ϊͭ���� ������ĸ��
A�������� B����չ�� C�������� D����������
��2������ˮ��ͷ����ʹ���������⣬������������� ��
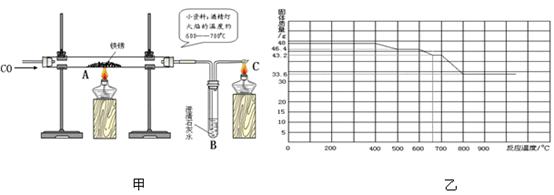
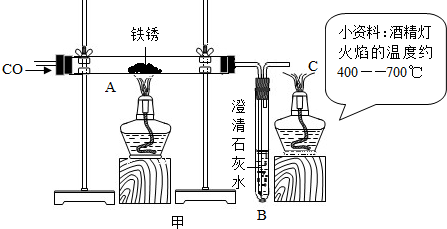
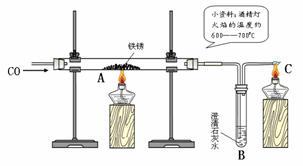
��3��С�����Լ�ˮ��ͷ��ȡ��һЩ������Ʒ������ͼ����ʾ��װ�ý���ʵ�顣
��C���ƾ��Ƶ������� ��ʵ�������С�������õĺ�ɫ�������ʷ��������������У�������������û������С���ô˽������ݵ������� ��
Ϊ������һ���������С���鵽�������ϣ�
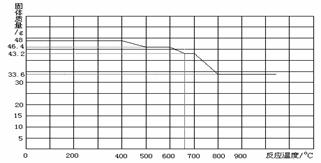
����֪������������FeO��Fe3O4��Fe2O3����һ�������£�������ʧȥ���е��������ձ���ԭΪ����
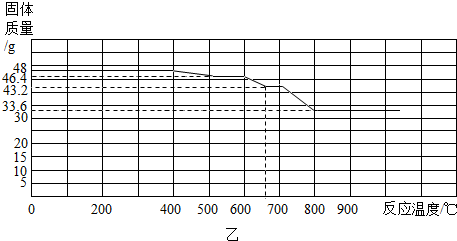
��ij����������������һ����̼�����ȷ�Ӧ���������������ݲ����Ƴ�ͼ�ҡ�

��ͨ����������֪����С��ʵ��ʧ�ܵ���Ҫԭ���� ����С����ʵ���У�A��������Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011�콭��ʡ�żҸ��г���5����Ӧ�Կ��Ի�ѧ�Ծ� ���ͣ������
�����й㷺Ӧ�ø��ֽ�����
��1��Ŀǰ������һ�����ϵ�ͭ���ڵ����͵��Ź�ҵ����Ҫ����Ϊͭ���� ������ĸ��
| A�������� | B����չ�� | C�������� | D���������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����й㷺Ӧ�ø��ֽ�����
��1��Ŀǰ������һ�����ϵ�ͭ���ڵ����͵��Ź�ҵ����Ҫ����Ϊͭ���� ������ĸ��
A�������� B����չ�� C�������� D����������
��2������ˮ��ͷ����ʹ���������⣬������������� ��
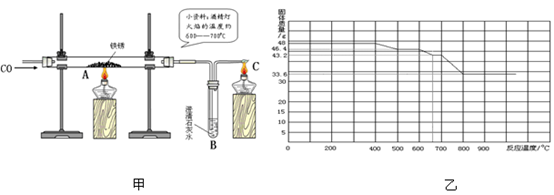
��3��С�����Լ�ˮ��ͷ��ȡ��һЩ������Ʒ������ͼ����ʾ��װ�ý���ʵ�顣
��C���ƾ��Ƶ������� ��ʵ�������С�������õĺ�ɫ�������ʷ��������������У�������������û������С���ô˽������ݵ������� ��
Ϊ������һ���������С���鵽�������ϣ�
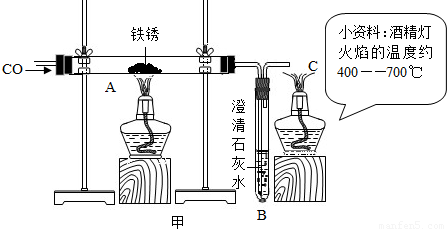
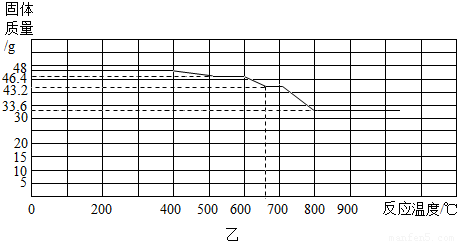
����֪������������FeO��Fe3O4��Fe2O3����һ�������£�������ʧȥ���е��������ձ���ԭΪ����
 ��ij����������������һ����̼�����ȷ�Ӧ���������������ݲ����Ƴ�ͼ�ҡ�
��ij����������������һ����̼�����ȷ�Ӧ���������������ݲ����Ƴ�ͼ�ҡ�
�� ��
��ͨ����������֪����С��ʵ��ʧ�ܵ���Ҫԭ���� ����С����ʵ���У�A��������Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011�걱����˳�����п���ѧһģ�Ծ��������棩 ���ͣ������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com