£Ø2012?ŹŠÖŠĒųČżÄ££©ČēĶ¼ĖłŹ¾ĪŖŹµŃéŹŅÖŠ³£¼ūµÄĘųĢåÖʱø”¢øÉŌļ”¢ŹÕ¼ÆµÄ²æ·ÖŅĒĘ÷£Ø×é×°ŹµŃé×°ÖĆŹ±£¬æÉÖŲø“Ń”ŌńŅĒĘ÷£©£®
ŹŌøł¾ŻĢāÄæŅŖĒ󣬻Ų“šĻĀĮŠĪŹĢā£ŗ£ØĢįŹ¾£ŗ3H
2+Fe
2O
32Fe+3H
2O£©
£Ø1£©ČōŌŚŹµŃéŹŅÖŠÓĆĻ”ŃĪĖįÓė“óĄķŹÆÖĘČ”²¢ŹÕ¼Æ“æ¾»”¢øÉŌļµÄ¶žŃõ»ÆĢ¼ĘųĢ壮£ØĢįŹ¾£ŗŃĪĖįÖŠ»Ó·¢³öµÄÉŁĮæHClĘųĢåæÉÓƱ„ŗĶĢ¼ĖįĒāÄĘČÜŅŗĪüŹÕ£©
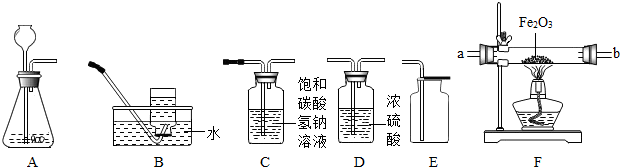
¢ŁĖłŃ”ŅĒĘ÷µÄĮ¬½ÓĖ³ŠņĪŖ
ACDE
ACDE
£ØĢīŅĒĘ÷ŠņŗÅ×ÖÄø£©£®
¢ŚÖĘČ”ĘųĢåµÄ»Æѧ·½³ĢŹ½ĪŖ
CaCO3+2HCl=CaCl2+H2O+CO2ӟ
CaCO3+2HCl=CaCl2+H2O+CO2ӟ
£®
¢ŪŹµŃéŹŅÖĘČ”¶žŃõ»ÆĢ¼ŗóµÄŹ£Óą·ĻŅŗÖŠ£¬³£ŗ¬ÓŠŹ£ÓąµÄÉŁĮæŃĪĖį£¬Óū¼ÓČė¹żĮæµÄŅ©Ę·³żČ„ŃĪĖį£¬µĆµ½½Ļ“æ¾»µÄÖŠŠŌŅŗĢ壬Ēė“ÓŹģŹÆ»Ņ”¢“æ¼ī”¢ŹÆ»ŅŹÆČżÖÖ¹ĢĢåŅ©Ę·ÖŠŃ”ÓĆŅ»ÖÖŹŌ¼Į½ųŠŠŹµŃ飬²¢¼ņŹö²Ł×÷·½·Ø
ŹÆ»ŅŹÆ£»¼ÓČė¹żĮæµÄŹÆ»ŅŹÆ£¬³ä·Ö·“Ó¦ŗó¹żĀĖ£¬¼“µĆµ½½Ļ“æ¾»µÄÖŠŠŌŅŗĢå
ŹÆ»ŅŹÆ£»¼ÓČė¹żĮæµÄŹÆ»ŅŹÆ£¬³ä·Ö·“Ó¦ŗó¹żĀĖ£¬¼“µĆµ½½Ļ“æ¾»µÄÖŠŠŌŅŗĢå
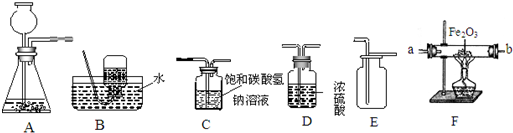
£Ø2£©ÓūÓĆŠæŗĶĻ”ĮņĖį·“Ó¦ÖĘČ”ĒāĘų£¬²¢²ā¶Øij²»“æŃõ»ÆĢśŃłĘ·ÖŠŃõ»ÆĢśµÄÖŹĮæ£ØŌÓÖŹ²»·“Ó¦£©£¬ĖłŃ”ŅĒĘ÷µÄĮ¬½ÓĖ³ŠņĪŖ£ŗA”śD”śF£®
¢ŁŹµŃ鏱£¬»į¹Ū²ģµ½×°ÖĆFÖŠµÄĻÖĻóĪŖ
ŗģÉ«±äŗŚÉ«£¬ŹŌ¹Ü±ŚÉĻÓŠĖ®Īķ
ŗģÉ«±äŗŚÉ«£¬ŹŌ¹Ü±ŚÉĻÓŠĖ®Īķ
£®
¢ŚČō·“Ó¦½ųŠŠĶźČ«ŗó£¬ŅĒĘ÷FµÄÖŹĮæ¼õÉŁa g£¬ŌņŃõ»ÆĢśŃłĘ·ÖŠµÄŃõ»ÆĢśµÄÖŹĮæĪŖ
g£Ø¼ĘĖć½į¹ūæÉĪŖ·ÖŹżŠĪŹ½£©£®
¢ŪŠæŗĶĻ”ĮņĖį·“Ó¦ŗó£¬ĖłµĆČÜŅŗÖŹĮæÓėÖ®Ē°Ļą±Č»į
A
A
£ØĢīŠ“ĻĀĮŠø÷ĻīŠņŗÅ£©
A£®Ōö“ó£»B£®¼õŠ”£»C£®²»±ä£»D£®ĪŽ·ØÅŠ¶Ļ£®









 2Fe+3CO2£©
2Fe+3CO2£©