
��9�֣�Сǿͬѧѡ������װ�ý���������ʵ�飬����ݲ�����գ�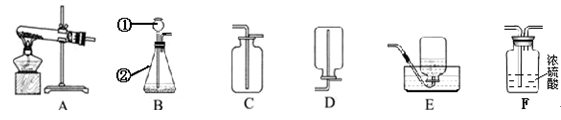
��1��д��ͼ����Ŷ�Ӧ���������ƣ��� ���� ��
��2���ø�����ع�����ȡ�����O2����ѡ�õ�װ������˳���� ��
��Ӧ�Ļ�ѧ����ʽ
��3��Сǿͬѧѡ�ù̡�Һ����ҩƷ������װ��BҲ������ȡ��������Ӧ�Ļ�ѧ����ʽΪ
��4��Сǿͬѧ����B��C��ȡ����һ�ֳ��������壬����Ϊ��ȡ�������� ��
��Ӧ�Ļ�ѧ����ʽ
��5�����Bװ�������ԵIJ����������ǣ�������Ƥ�����Ӻõ����ܣ�Ȼ����Ӽ�ס��Ƥ�ܣ�������м���ˮ���γ�һ��ˮ�������ã����۲쵽___ ��˵�����������á�

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ͨ��һ��Ļ�ѧѧϰ�����Ѿ�������ʵ������ȡ������йع��ɡ�������ͼ�ṩ��һЩʵ��װ�ûش����⣺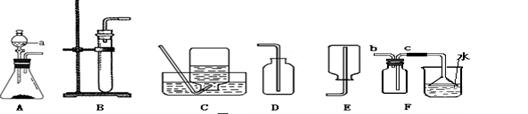
��1������Aװ����ȡ����������a������� �������Ļ�ѧ����ʽΪ �� ��
��2��ͨ���������ϵ�֪������(NH3)��һ���ܶȱȿ���С�Ҽ�������ˮ�����壬��ˮ��Һ��Ϊ��ˮ������ѡ�������Ȼ�狀��������ƵĹ�����������ȡ����ʱ����ѡ��Bװ������ɸ�ʵ�飬����еĸĶ��� ��
��3������Ϊ��ѡ���ռ�������װ��������� ������ţ���ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��8�֣�ʵ������������ء��������̡�ʯ��ʯ��ϡ���ᣬ�Լ�����������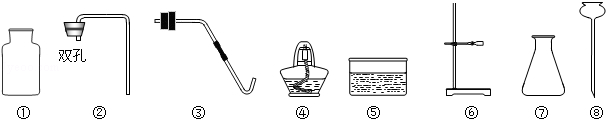
��1��д��ͼ������5��������_________����
��2����������������ҩƷ������ȡ���ռ�������̼����ѡ�����������_________��������ţ�������Ӧ�Ļ�ѧ����ʽΪ��_________������2�֣�
��3��������һ��������_________��������������װ�ú�ҩƷ��������ȡ���ռ���������ѡ�����������_________��������ţ�����Ӧ�Ļ�ѧ����ʽ����_________������2�֣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ʵ������������������
A��O2��2KClO3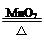 2KCl��3O2��
2KCl��3O2��
B��CO2��
C��Cl2��MnO2��4HCl(Ũ) MnCl2��Cl2����2H2O
MnCl2��Cl2����2H2O
˵����Cl2��һ�ֻ���ɫ���д̼�����ζ���ܶȱȿ�����������ˮ�����塣
д����CO2�ķ�Ӧ����ʽ
��������������װ��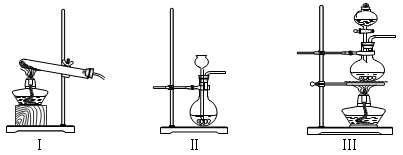
���ּ���װ��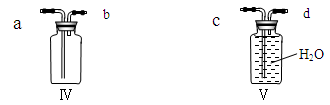
��ȡCO2Ӧѡȡ�ķ���װ���� ��Ӧѡ�â�װ����ȡ�������� ��
�â�װ���ռ�Cl2Ӧ�� �ڽ������������������У����â�װ���ռ��������� ��Ӧ�� �ڽ�����
A����O2��ʣ������з����MnO2�IJ����� �������ɣ������õ��IJ��������� ��
��100g 36��5%��Ũ����������MnO2��ϼ��ȣ����������Ƶ�Cl2������Ϊ g����ʵ���ռ�����Cl2����ԶԶС�������ֵ����������ȷ��װ�ò�©��������ܵ�ԭ����
, (�����)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��5�֣���ʵ������ȡ��������װ������ͼ��ʾ�� 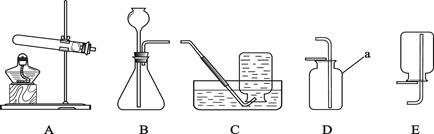
��ش��������⣺
��1��װ���бꡰa��������������_________________��
��2��ʵ�����ø��������ȡ������ѡ�õ��ռ�װ����_______������ĸ��ţ��� ��Ӧ�Ļ�ѧ����ʽΪ ��
��3��ʵ�����ô���ʯ��ȡ������̼��ѡ�õķ���װ����_________����������Ϊ _��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ��ʾʵ��װ�ó�������ȡ������������̼�����������壮��ش����⣺
��1��д�������ٵ�������_________����
��2������BCװ�������ȡһ�����壬�䷢����Ӧ�Ļ�ѧ����ʽ����_________����
��3������AEװ�������ȡһ�����壬�䷢����Ӧ�Ļ�ѧ����ʽ����_________����
��4����֪��Ȳ�����ܶȱȿ�����С�����ɿ�״̼���ƣ�CaC2����ˮ��Ӧ�Ƶã�ͬʱ����һ����ʯ�ҹ��壬ʵ��������ȡ���ռ���������Ȳ��ѡ��ͼ�е�װ������_________��������ţ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��10�֣����и�ͼΪ���������Ʊ������ʺ���ʵ��IJ�����������ѡ��������ɺ��ʵ�ʵ��װ������������⣺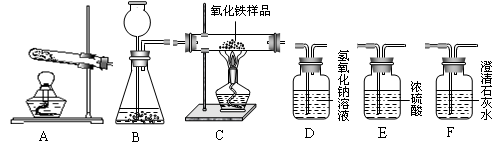
��1��ʵ���ҳ���п����ϡ���ᷴӦ��ȡ��������ѧ����ʽΪ ��
����ȡ�������������ԭ��������Ʒ�����ⶨ��������Ʒ�Ĵ��ȣ���ѡװ�����ӵ�˳��Ϊ��
װ�â� ��װ�â� ��װ�â� ����д���������ĸ����
���ȥ��װ�âڣ��Բⶨ��� ���л��ޣ�Ӱ�죿
��2���ƾ�����������������ȼ�գ�����һ����̼��������̼��ˮ�Ļ�����塣ij��ȤС���ͬѧ��ͨ��ʵ��ⶨ�Ҵ�����������������ȼ����������һ����̼���ⶨ��������Ʒ�Ĵ��ȣ���������װ�õ�˳��Ϊ���������ͨ��װ��D��װ��E��װ��C��װ��F����ش𣬢�װ��D��������
��װ��F�е�������
�������װ��D��װ��E����λ�ã��Բⶨ��� ���л��ޣ�Ӱ�죿
�ܸ���װ������в���֮����������Ʋ��䷽��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��11�֣�������ͼ�ش����⣺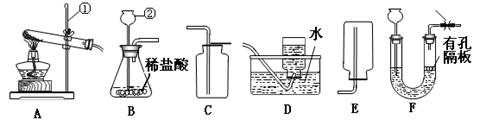
��1����Ţ٢ڵ��������ƣ��� ���� ��
��2��ʵ����ѡ��A��Dװ����ȡO2�������ռ����ʱ��Ӧ�� �����ˮ��ȡ�������ܡ���Ϩ��ƾ��ơ�����
��3��ʵ���ҿ����Ȼ�粒������ʯ�һ�ϼ�����ȡNH3����Ӧ����ʽΪ
��4����ѡ����װ��B���ռ�װ�� ��ѡ����ţ���������ʵ������ȡCO2����������װ����B��ΪF�����ŵ��� ��
��Ϊ�˵õ������������CO2������װ�ã�����ͼ���ĵ��ܰ�������������˳���� ��ѡ����ĸ����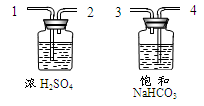
a��1��2��4��3 b��4��3��1��2
c��2��1��3��4 d��3��4��2��1
���û�ѧ��Ӧ����ʽ��ʾ����NaHCO3��Һ������ ��
��5����������ͼ��ʾװ����ȡ�������ⶨ�������ʵ��ǰӦ���װ�õ������ԣ�Ϊ��ȷ�ⶨ����������������������ʱӦ��ע��������������������������ȴ�����£��ڳ���©��������б����Ӧ�����ƶ�ˮ�ܣ�������Һ����ƽ�ٶ�����������Ӧ��Һ�尼Һ����ʹ�����ˮƽ������Ϊ����Ҫ���������� ��������ţ���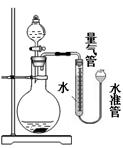
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ��ʵ������ȡ����ʱ������װ�ã�����G��IΪ����װ�ã�������װ�ûش��������⣺
��1�������ٵ����� ��
��2����Aװ����ȡ��ij�����ʹ�����ǵ�ľ����ȼ����д����Aװ����ȡ������Ļ�ѧ����ʽ ��
��3������Gװ���ռ���һƿ��ɫ��ζ�����壬���������Ƿ�ΪCO2�ķ����� ��������Fװ���ռ������壬������Ӧ�� ���a����b����ͨ�룮
��4�������£�������NH3����һ���д̼�����ζ�����壬��������ˮ���ܶȱȿ���С����Һ�ʼ��ԣ�ʵ������ȡ������ԭ����NH4C1���̣���Ca��OH��2���̣���Ϲ��ȣ���Ҫ�Ƶø���İ���������β������ֹ��Ⱦ��������ѡ���װ������˳��������B�� ��F��H����װ����ţ���
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com