
| ̼���Ƶ����� |
| ���������� |
| 106 |
| 197 |
| x |
| ng |
| 106n |
| 197 |
| ||
| mg |
| 106n |
| 197m |
| 106n |
| 197m |


 ���Ž�����ٰθ��νӹ㶫���������ϵ�д�
���Ž�����ٰθ��νӹ㶫���������ϵ�д� �����������ҵ�������������ϵ�д�
�����������ҵ�������������ϵ�д� ѧ���������ν��Ͼ���ѧ������ϵ�д�
ѧ���������ν��Ͼ���ѧ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ���� | װ�â� | װ�â� | |
| ��Ӧǰ������/g | 15.8 | 182.3 | 212.2 |
| ��Ӧ�������/g | 14.4 | 184.1 | 216.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������

| ���� | װ�â� | װ�â� | |
| ��Ӧǰ������/g | 15.8 | 182.3 | 212.2 |
| ��Ӧ�������/g | 14.4 | 184.1 | 216.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
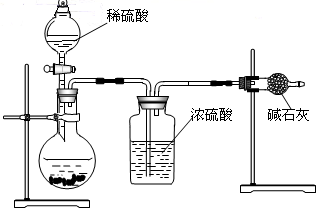
ij��ѧ��ȤС��ͬѧΪ�˲ⶨ������̼��������Ԫ�ص������ȣ��������ͼ��ʾ��ʵ�顣ʵ�鲽�����£��ȳ�������װ�â��װ�â����������ͼʾ����װ�ã���ȼ����һ��ʱ�������Ϩ���ٷֱ��������װ�â��װ�â��������ʵ���������±���

���� | װ�â� | װ�â� | |
��Ӧǰ������/g | 15.8 | 182.3 | 212.2 |
��Ӧ�������/g | 14.4 | 184.1 | 216.6 |
�������ͼװ�ú����ݻش����⣺
��1��װ�â���Ũ����������� ��
װ�â����������ƺ������ƹ���������� ��
��2�����ϱ����ݿ�֪����Ӧ������ˮ �ˣ�������̼ �ˡ�
��3���ɸ�ʵ�����ݼ��㣬������̼Ԫ�غ���Ԫ�ص�������Ϊ ��
��4��װ�â��װ�â��ܷ�Ե��� ����ܡ����ܡ�����ԭ����
��5����С��ͬѧ����װ�â��װ�â����ӵ�����������������ٵ���������������ѧ����֪ʶ�������ǵ��ɻ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2008���Ϻ��мζ����п���ѧ��ģ�Ծ��������棩 ���ͣ������

| ���� | װ�â� | װ�â� | |
| ��Ӧǰ������/g | 15.8 | 182.3 | 212.2 |
| ��Ӧ�������/g | 14.4 | 184.1 | 216.6 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com