
ij��������к���������һ����̼��������̼��ˮ������Ϊ����ʵ��֤���û�������к������������ʣ����ṩ����װ����ͼ��ʾ���ش��������⣺
����
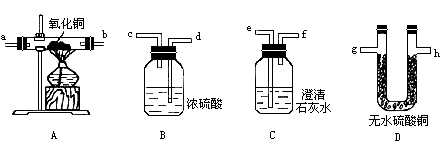
������������װ�õ���ȷ˳����(��д���ӿ���ĸ��ÿ��װ�����������ã��ɸ�����Ҫѡ���Σ����ҽӿ���Ų���)��


��Aװ������________�Σ�������Ӧ�Ļ�ѧ����ʽΪ_______________��
��Bװ������________�Σ�ÿ�ε�������________________��
��Cװ������________�Σ�ÿ�ε�������________________��
��Dװ������________�Σ�ÿ�ε�������________________��
��β���ܷ��ŷŵ�������ȥ��Ϊʲô��Ӧ����������
____________________________________________________
____________________________________________________��
|
�����ٻ������(g)(h)��(e)(f)��(d)(c)��(a)(b)��(g)(h)��(e)(f)��β�� ������1, ������1,����ˮ���������õ������CO�� ������2,��һ����֤������ȥ������е� ������2,��һ����֤������ȥ������е�ˮ�������ڶ�����֤��������к��� ���������ŷŵ������У���Ϊβ���к��е�CO�ж�����Ⱦ����������β��ͨ���ƾ��ƻ����ȼ��ʹCOת��Ϊ |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����


| ʵ����� | ʵ������ | ʵ����� | |
| �� | �ڼ��촦��ȼ���壬 �ڻ����Ϸ���һ�� ������ྻ���ձ� |
�ձ��ڱ� ������ɫҺ�� |
�������������� �� |
| �� | �ڻ����Ϸ���һ��Ϳ ��������ʯ��ˮ���ձ� |
�ձ��ڳ��� ��ʯ��ˮ����� |
��������к���һ����̼ �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

 [ʵ�����]������ȷ��
[ʵ�����]������ȷ��| ʵ�鲽�� | ʵ������ | ʵ����� | |
| �٣�19�� | �ڼ��쵼�ܴ���ȼ���壬 �ڼ��쵼�ܴ���ȼ���壬�ڻ����Ϸ���һֻ������ձ� �ڼ��쵼�ܴ���ȼ���壬�ڻ����Ϸ���һֻ������ձ� �� |
�ձ��ڱ���ˮ�� �ձ��ڱ���ˮ�� |
ԭ��������������� ԭ��������������� �� |
| �ڣ�20�� | �������ձ���Ѹ�ٵ�����������ʯ��ˮ�������ڻ����Ϸ���һֻ�ڱ�Ϳ�г���ʯ��ˮ���ձ����� �������ձ���Ѹ�ٵ�����������ʯ��ˮ�������ڻ����Ϸ���һֻ�ڱ�Ϳ�г���ʯ��ˮ���ձ����� �� |
����ʯ��ˮ����ǣ���������ɫ���� ����ʯ��ˮ����ǣ���������ɫ���� |
ԭ�����������һ����̼ ԭ�����������һ����̼ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij��ѧ��ȤС���ˮ����ͨ�����ȵ�ľ̿�õ��Ļ���������Ҫ�ɷֲ�������Ȥ��ͬѧ�Ǿ���ͨ��ʵ�����̽����
��������롿�û��������Ҫ�ɷ�Ϊһ����̼��������̼��������ˮ������
���������ϡ�a��Ũ�������������� b�������ڼ��ȵ�������������ͭ��![]() Ӧ����ͭ��ˮ��
Ӧ����ͭ��ˮ��
��ʵ����̡�ͬѧ������ʦ��ָ�������������ͼ��ʾװ�ã���������ʵ��(���ּг���������ȥ)��������ÿ��װ�����ҩƷ��������Ӧ��֣�
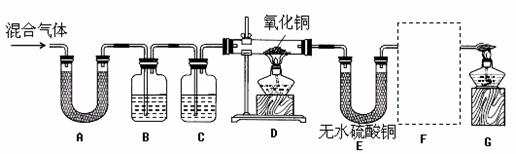
��1��װ��A����ˮ����ͭ������װ��B�г���ʯ��ˮ����ǡ��ɴ˵ó���������к�
�� ���塣д��װ��B�з�Ӧ�Ļ�ѧ����ʽ ��
��2��ͬѧ��ͨ���۲�װ��D��E�е������ ��Ϊ��ȷ�ϻ�������к�������������ȷװ��C���Լ������Ƽ���;�� ��Ϊ��֤������������Ĵ��ڣ�װ��F�е�ҩƷ������ͽ���![]() �� ��
�� ��
��ʵ����ۡ�������ȷ��
��ʵ�鷴˼���������ۣ�ͬѧ�ǽ���ͼ��װ�ý����˼��Ľ����װ������ͼ��ʾ��
��3��д��U��A�з�����Ӧ�Ļ�ѧ����ʽ ��
| ʵ�鲽�� | ʵ������ | ʵ����� | |
| �٣�19�� | �ڼ��쵼�ܴ���ȼ���壬 �� |
| �� |
| �ڣ�20�� | ��
|
|
|
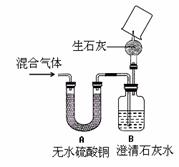 ��4��ͬѧ��Ϊ����֤ͨ������ܺ�����ijɷ֣���������
��4��ͬѧ��Ϊ����֤ͨ������ܺ�����ijɷ֣���������![]() ��ʵ��Ϊ��
��ʵ��Ϊ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012���Ϻ��г������п���ѧһģ�Ծ��������棩 ���ͣ������
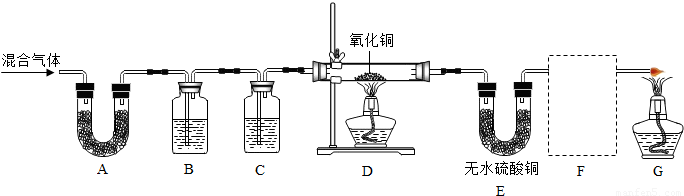
| ʵ�鲽�� | ʵ������ | ʵ����� | |
| �٣�19�� | �ڼ��쵼�ܴ���ȼ���壬______�� | ______ | ______�� |
| �ڣ�20�� | ______�� | ______ | ______ |
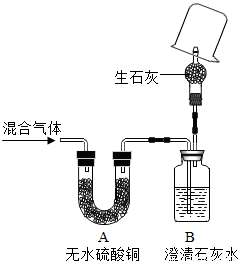
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com