
Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ������������������������ij��ѧ��ȤС���ͬѧΪ�˲ⶨij��ͭ����ɣ�ȡ20g�û�ͭ��Ʒ���ձ��У������з�5�μ�����ͬ��������������ϡ���ᣬʹ֮��ַ�Ӧ��ÿ������ϡ�����������ʣ������������¼���±���
| | ����ϡ�����������g�� | ��ַ�Ӧ��ʣ������������g�� |
| ��1�� | 20 | 17.4 |
| ��2�� | 20 | 14.8 |
| ��3�� | 20 | 12.2 |
| ��4�� | 20 | 12.0 |
| ��5�� | 20 | m |
��1��12 ��1�֣� ��2��40% ��2�֣���3��19.6%
���������������1���ɱ������ݿ�֪��ÿ����20gϡ���ᣬ������Ӧ2.6gп������4�ε�ʱ����ֻ������0.2g��˵����ʱп�Ѿ�ȫ����Ӧ���ˣ��ʵ�5�μ������ᣬ������������ı䣬����m=12g��
��2����ͭ��Ʒ��п����������Ϊ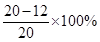 =40%��
=40%��
��3����20gϡ������ȫ��Ӧ��п������ = 20g-17.4g=2.6g
�⣺�裬20gϡ������H2SO4������Ϊx��
Zn + H2SO4 = ZnSO4 + H2��
65 98
2.6g x 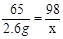
x=3.92g
����ϡ���������ʵ���������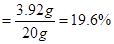
���㣺��Һ�����ʵ��������������ݻ�ѧ�����Ǽ��㡣
���������ݻ�ѧ����ʽ���㣬Ҫע�����IJ��裬�衢д���ҡ��С��⡢��
������������= ��
��


 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ��һ�� | �ڶ��� | ������ | |
| ����ϡ�����������g�� | 50 | 50 | 50 |
| ����������������g�� | 0.4 | m | 0.2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��2013?�ֿ���һģ��Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ��������������������
��2013?�ֿ���һģ��Cu��Zn�ĺϽ��Ϊ��ͭ���������ĵ����Ժ���ʴ�ԣ��������������������| �Ͻ������/g | ϡ��������/mL | �������������/mL | |
| ��һ�� | 2 | 15 | 44.4 |
| �ڶ��� | 2 | 20 | 44.4 |
| ������ | 4 | 15 | 44.4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ����ϡ�����������g�� | ��ַ�Ӧ��ʣ������������g�� | |
| ��1�� | 40 | m |
| ��2�� | 40 | 14.8 |
| ��3�� | 40 | 12.2 |
| ��4�� | 40 | 11.2 |
| ��5�� | 40 | 11.2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| �Ͻ������/g | ϡ��������/mL | ��������������/g | |
| ��1�� | 2 | 15 | 0.04 |
| ��2�� | 2 | 20 | 0.04 |
| ��3�� | 4 | 15 | 0.04 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com