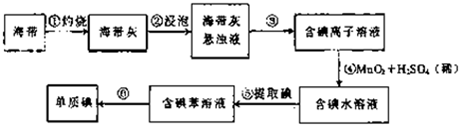
ij�о���ѧϰС����Ʋ�����������ʵ�飮
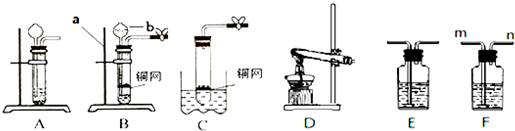
��1������ͬѧ��ͼװ����ȡ���﴿���Ķ�����̼���壮��ش��������⣺
������X��������
��ʵ��ǰ���رջ�������X�м���ˮʹX��Һ������Թ��е�Һ�棬һ��ʱ���Һ�汣�ֲ���˵��
��
��Ϊ��ȥCO
2�л��е�HCl���壬Bװ����ʢ�ű���NaHCO
3��Һ��д���÷�Ӧ�Ļ�ѧ����ʽ
��
��Ҫ�õ������CO
2��Cװ��Ӧʢ��
�����Լ����ƣ���
����˵��װ��B��C���Լ����ܽ�����ԭ��
��
��2������ͬѧΧ��CO��ԭCuO�����ĺ�ɫ�����Ƿ�һ����Cu����̽����
�������ϣ�
��Cu
2O��ĩ�ʺ�ɫ����������Һ�У�Cu
2O+H
2SO
4=Cu+CuSO
4+H
2O
���ڿ����и�������ʱ��Cu
2O�ȶ�����CuO���ֽ�����Cu
2O��O
2������룺
�����ɫ������Cu��
�����ɫ������Cu
2O��
�����ɫ������
��
ʵ��̽����
�����ȡһ�������ĺ�ɫ����a g���ڿ����и������գ�ֱ���������ٷ����ı䣬�������ù�������Ϊb g��
��������ݷ���
����a=b�������
������
����b��a����˵����ɫ������һ������
���ʣ�
�������ȡ������ɫ����Ͷ��ϡ�����У���ַ�Ӧ����Һ����ɫ�����ݴ����ú�ɫ�����к���
�����Cu����Cu
2O�Ļ�ѧ����ʽ��
��


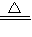 2Cu2O��
2Cu2O��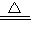 2Cu2O
2Cu2O

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
 ��2012?�紨��һģ��ij�о���ѧϰС����Ʋ�����������ʵ�飮
��2012?�紨��һģ��ij�о���ѧϰС����Ʋ�����������ʵ�飮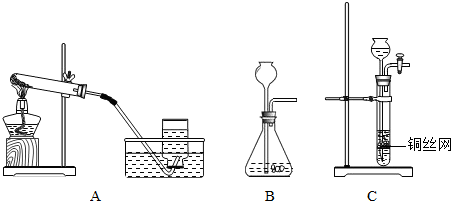
 ij�о���ѧϰС����Ƶ�ʵ��װ�ã���ͼ�����ȿ���ȡ���壬�ֿ�������֤�������ʣ�
ij�о���ѧϰС����Ƶ�ʵ��װ�ã���ͼ�����ȿ���ȡ���壬�ֿ�������֤�������ʣ�