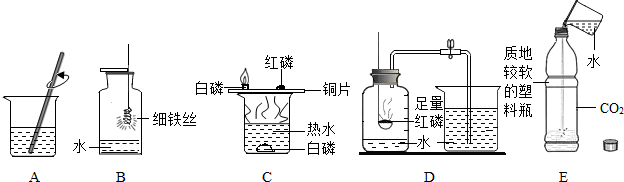
��2013?������һģ������������ǻ�ѧ��ȤС���Ա�����������µķ�����ȡ������
���������������ϵõ���
���ò�ľ���к��е�̼��ؿ�����ȡ�������أ�
������������Һ��������Ӧ��ȡ������
���Ǵ�ֲ��ȼ�պ�IJ��������ռ���ľ�ҽ�������ʵ�飺
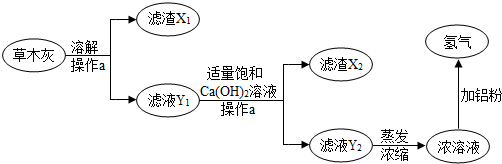
��1������a��������
����
����
������a���õ��IJ����������˲�������©��������Ҫ
�ձ�
�ձ�
������X
2�ɷ���
CaCO3
CaCO3
���ѧʽ����
��2������ľ���ܽ�ʱ��Ӧ�þ������ٵ�ˮ���ҽ��ݹ�ҹ��Ŀ����
�ò�ľ���е�K2CO3����ܽ⣬������Һ�����ܴﵽ����
�ò�ľ���е�K2CO3����ܽ⣬������Һ�����ܴﵽ����
��
��3������ҺY
2��Ũ��Һ�мӷ���м��ˮҲ�Ƿ�Ӧ�����������ƫ����أ�KAlO
2�����÷�Ӧ�Ļ�ѧ����ʽ��
2Al+2KOH+2H2O=2KAlO2+3H2��
2Al+2KOH+2H2O=2KAlO2+3H2��
���÷�Ӧ�У�
�⡢��
�⡢��
Ԫ�صĻ��ϼ��ڷ�Ӧǰ���б仯��



 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
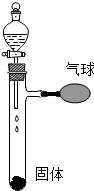 ��2013?������һģ��������ͼ��ʾ����Һ©����ʹ���е���ɫҺ�壨���������Թ��еĹ��壨�������Ӵ���Ӧ���ɹ۲쵽�����������ʹ��������ֱ�д��һ������ͼ�����������Ҫ��Ļ�ѧ����ʽ��
��2013?������һģ��������ͼ��ʾ����Һ©����ʹ���е���ɫҺ�壨���������Թ��еĹ��壨�������Ӵ���Ӧ���ɹ۲쵽�����������ʹ��������ֱ�д��һ������ͼ�����������Ҫ��Ļ�ѧ����ʽ�� ��2013?������һģ��A��B��C��D��E ���dz��л�ѧ���������ʣ�����AΪ���Σ�B��D��Ϊ����������Ԫ�ص����壬X��Y��Ϊ��ɫ��ĩ��FΪ��ɫ�������ʣ�E�ڳ�����ΪҺ̬������֮���ת����ϵ����ͼ��ʾ����Ӧ�����Ͳ��ֲ�������ȥ�����Իش��������⣮
��2013?������һģ��A��B��C��D��E ���dz��л�ѧ���������ʣ�����AΪ���Σ�B��D��Ϊ����������Ԫ�ص����壬X��Y��Ϊ��ɫ��ĩ��FΪ��ɫ�������ʣ�E�ڳ�����ΪҺ̬������֮���ת����ϵ����ͼ��ʾ����Ӧ�����Ͳ��ֲ�������ȥ�����Իش��������⣮