”¾“š°ø”æ
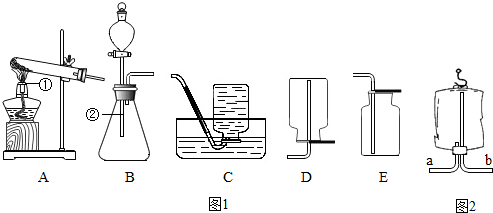
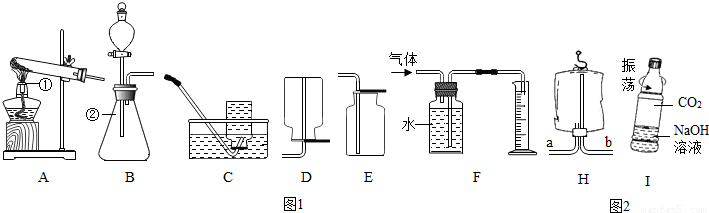
·ÖĪö£ŗ£Ø1£©ŹģĮ·ÕĘĪÕ³£¼ū»ÆѧŅĒĘ÷µÄĆū³Ę¼°ĘäÓĆĶ¾£»
£Ø2£©¼ÓČČĀČĖį¼ŲŗĶ¶žŃõ»ÆĆĢµÄ»ģŗĻĪļÖĘČ”ŃõĘų£¬·¢Éś×°ÖƵÄĢŲµćŹōÓŚ¹ĢĢå¼ÓČČŠĶ£»øł¾ŻŃõĘųµÄĆܶČŗĶČܽāŠŌŃ”ŌńŹÕ¼Æ·½·Ø£¬ÅÅæÕĘų·ØŹÕ¼ÆµÄĘųĢå½ĻøÉŌļ£¬ÅÅĖ®·ØŹÕ¼ÆµÄŃõĘų½Ļ“æ¾»£»
£Ø3£©ŹµŃéŹŅÓĆH
2O
2ČÜŅŗŗĶMnO
2»ģŗĻÖĘŃõĘų£¬¶žŃõ»ÆĆĢŹĒ“߻ƼĮ£¬øıäĮĖ¹żŃõ»ÆĒāČÜŅŗµÄ·Ö½āĖŁĀŹ£»øł¾ŻŹµŃéµÄ½į¹ūĶĘ²āŌŅņ£»
£Ø4£©øł¾ŻĒāĘųµÄĆܶČŃ”Ōń½ųĘųæŚ£»
£Ø5£©øł¾ŻĖ®ŗĶĒāŃõ»ÆÄĘČÜŅŗ¶Ō¶žŃõ»ÆĢ¼ĪüŹÕÄÜĮ¦µÄ²»Ķ¬»Ų“š£¬¶žŃõ»ÆĢ¼ŗĶĒāŃõ»ÆÄĘ·“Ó¦²śÉśĢ¼ĖįÄĘŗĶĖ®£®
½ā“š£ŗ½ā£ŗ£Ø1£©¢ŁŹĒ¾Ę¾«µĘ£» ¢ŚŹĒ׶ŠĪĘ棻
£Ø2£©¼ÓČČĀČĖį¼ŲŗĶ¶žŃõ»ÆĆĢµÄ»ģŗĻĪļÖĘČ”ŃõĘų£¬·¢Éś×°ÖƵÄĢŲµćŹōÓŚ¹ĢĢå¼ÓČČŠĶµÄA×°ÖĆ£»
ŅņĪŖŃõĘų²»Ņ×ČÜÓŚĖ®ĒŅĆܶȓóÓŚæÕĘų£¬ĖłŅŌ¼ČæÉŅŌÓĆÅÅĖ®·ØŹÕ¼Æ£¬Ņ²æÉŅŌÓĆĻņÉĻÅÅæÕĘų·ØŹÕ¼Æ£»ÅÅĖ®·ØŹÕ¼ÆµÄĘųĢå½Ļ“棬¶ųĻņÉĻÅÅæÕĘų·ØŹÕ¼ÆµÄĘųĢå½ĻøÉŌļ£¬¹ŹÓ¦Ń”ŌńµÄŹÕ¼Æ×°ÖĆŹĒE£»
£Ø3£©ŹµŃéŹŅÓĆH
2O
2ČÜŅŗŗĶMnO
2»ģŗĻÖĘŃõĘų£¬¶žŃõ»ÆĆĢŹĒ“߻ƼĮ£¬øıäĮĖ¹żŃõ»ÆĒāČÜŅŗµÄ·Ö½āĖŁĀŹ£¬Ęšµ½“ß»Æ×÷ÓĆ£»øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ£ŗ2H
2O
2 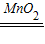
2H
2O+O
2”ü£»
ĶعżÅÅĖ®ĮæĄ“²ā¶ØÉś³ÉŃõĘųµÄĢå»ż£¬·“Ó¦½įŹųŗ󣬷¢ĻÖĮæĶ²ÄŚŹÕ¼Æµ½µÄĖ®µÄĢå»ż×ÜŹĒ±ČĄķĀŪֵʫ“ó£ØĖ®µÄĢå»ż²āĮæ×¼Č·£©£¬ĘäÖ÷ŅŖŌŅņæÉÄÜŹĒ£ŗ“Ó·ÖŅŗĀ©¶·Į÷ČėµÄĖ«ŃõĖ®Õ¼ÓŠŅ»¶ØµÄĢå»ż£Ø»ņ·“Ó¦·ÅČČ£¬ĘųĢåÅņÕĶ£©£»
£Ø4£©ŅņĪŖĒāĘųµÄĆܶȱČæÕĘųŠ”£¬¹ŹĒāĘų“Ó³¤¹Ü½ųČėµ½Ņ½ÓĆĖÜĮĻ“üµÄÉĻ·½£¬°ŃæÕĘų“Ó¶Ģ¹ÜÅųö£»
£Ø5£©æóČŖĖ®Ęæ±ä±ā£¬æÉŅŌŹĒ¶žŃõ»ÆĢ¼ČÜÓŚĖ®Ź¹ĘæÄŚµÄĘųŃ¹±äŠ”£¬Ņ²æÉŅŌŹĒ¶žŃõ»ÆĢ¼ÓėĒāŃõ»ÆÄĘ·“Ó¦Ź¹ĘæÄŚµÄĘųŃ¹±äŠ”µÄ½į¹ū£¬ĖłŅŌŅŖÖ¤Ć÷CO
2ÓėNaOHČÜŅŗČ·Źµ·¢ÉśĮĖ·“Ó¦£¬æÉŅŌĶعż¶ŌĖÜĮĻĘæ±ä±āµÄ³Ģ¶Č¶Ō±ČĄ“Ķź³É£ŗĒāŃõ»ÆÄĘČÜŅŗÖŠ¼ČÓŠĖ®Óė¶žŃõ»ÆĢ¼·“Ó¦£¬ÓÖÓŠĒāŃõ»ÆÄĘŗĶĖ®·“Ó¦£¬Ņņ“ĖĖÜĮĻĘæ»į±äµÄøü±ā£»CO
2ÓėNaOHČÜŅŗ·“Ó¦²śÉśĢ¼ĖįÄĘŗĶĖ®£¬·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ£ŗCO
2+2NaOH=Na
2CO
3+H
2O£®
¹Ź“š°øĪŖ£ŗ£Ø1£©¢Ł¾Ę¾«µĘ£» ¢Ś×¶ŠĪĘ棻 £Ø2£©A”¢E£»£Ø3£©“߻ƣ¬2H
2O
2 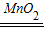
2 H
2O+O
2”ü£»“Ó·ÖŅŗĀ©¶·Į÷ČėµÄĖ«ŃõĖ®Õ¼ÓŠŅ»¶ØµÄĢå»ż£ØČōĢī·“Ó¦·ÅČČ£¬ĘųĢåÅņÕĶŅ²æÉ£©
£Ø4£©b£»£Ø5£©½«ĒāŃõ»ÆÄĘČÜŅŗČÜŅŗ»»³ÉµČĢå»żµÄÕōĮóĖ®×÷¶Ō±ČŹŌŃ飻CO
2+2NaOH=Na
2CO
3+H
2O£®
µćĘĄ£ŗ±¾Ģāæ¼²éĮĖ³£¼ūĘųĢåŃõĘųµÄÖĘČ”×°ÖĆ”¢ŹÕ¼Æ·½·ØµÄŃ”Ōń¼°ŹµŃéĪó²īµÄ·ÖĪö”¢¶žŃõ»ÆĢ¼µÄŠŌÖŹĢ½¾æ£¬æÉŅŌøł¾ŻÖĘČ”ĘųĢå·¢Éś×°ÖĆ”¢ŹÕ¼Æ×°ÖĆŃ”ŌńµÄŅĄ¾Ż½įŗĻŹµŃéµÄĻÖĻóŗĶ½įĀŪÖšŅ»·ÖĪö£®

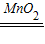 2H2O+O2”ü£»
2H2O+O2”ü£»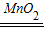 2 H2O+O2”ü£»“Ó·ÖŅŗĀ©¶·Į÷ČėµÄĖ«ŃõĖ®Õ¼ÓŠŅ»¶ØµÄĢå»ż£ØČōĢī·“Ó¦·ÅČČ£¬ĘųĢåÅņÕĶŅ²æÉ£©
2 H2O+O2”ü£»“Ó·ÖŅŗĀ©¶·Į÷ČėµÄĖ«ŃõĖ®Õ¼ÓŠŅ»¶ØµÄĢå»ż£ØČōĢī·“Ó¦·ÅČČ£¬ĘųĢåÅņÕĶŅ²æÉ£©

 Č«ÄÜĮ·æ¼¾ķĻµĮŠ“š°ø
Č«ÄÜĮ·æ¼¾ķĻµĮŠ“š°ø