
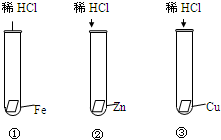
 ijͬѧΪ̽��Fe��Zn��Cu���ֽ����Ļ��˳����������ʵ�飺
ijͬѧΪ̽��Fe��Zn��Cu���ֽ����Ļ��˳����������ʵ�飺

 ������ȫ��������ϵ�д�
������ȫ��������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij��ȤС���ͬѧ̽��Ũ������������̼�ظ֣���Ӧ���������ijɷ֣�
���������ϡ���Fe��ŨH2SO4��Ӧ�����ȣ���SO2����
��C��ŨH2SO4��Ӧ�����ȣ���CO2��SO2����
��SO2��ʹ����ʯ��ˮ�����
��SO2��ʹƷ����Һ�ĺ�ɫ��ȥ����CO2����
��SO2��������KMnO4��Һ��Ӧʹ����ɫ����CO2����
��ʯ�ҵ���Ҫ�ɷ�ΪNaOH��Cao
��ʵ��̽����
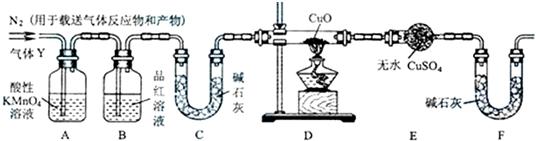
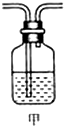
��1����ȡ24.0g��������60.0mLŨ�����У����ȣ���ַ�Ӧ��õ�����ҺX���ռ�������Y����ͬѧͨ��ʵ��ⶨ����֪����Y��SO2������������Ϊ66.7%��ͬѧ����Ϊ����Y�л����ܻ���H2��Z���壬��Z���������_______��̽��ʵ�����Ҫװ����ͼ��ʾ��

��2��װ��A���Լ���������____________��
��3��Ϊȷ��Z�Ĵ��ڣ���Ҫ��װ�üף���ͼ��ʾ����������װ�õģ����ţ� ֮�䣮װ�ü��е�Һ�������� ��

��4��ʵ���У�����۲쵽װ��D�� ����װ��E�� �����ȷ��Y�л���H2���壮
��5�������Ҫ�ⶨ���������Y��H2�ĺ�����H2ԼΪ0.01g���������ò���H2��������⣬ ��ѡ����ܡ����ܡ����ó�����ͼ��װ��D��װ��E�������仯�ķ�����ͨ������˵�����жϵ����ɣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011�꽭��ʡ�Ͼ����п���ѧ�Ծ��������棩 ���ͣ������


�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com