
”°µĶĢ¼³ĒŹŠ”±µÄĄķÄī¹į“©ŌŚÉĻŗ£ŹĄ²©Ō°µÄ½ØÉčÖŠ”£
£Ø1£©”°µĶĢ¼³ĒŹŠ”±µÄ½ØÉč¼õÉŁĮĖ¶žŃõ»ÆĢ¼ĘųĢåµÄÅÅ·Å£¬Äܼõ»ŗ µÄ³Ģ¶Č£»×ŌČ»½ēÖŠĻūŗĶžŃõ»ÆĢ¼µÄÖ÷ŅŖĶ¾¾¶ŹĒ £»ĒėÄ抓³öŅ»ÖÖČÕ³£Éś»īÖŠ·ūŗĻ”°µĶĢ¼¾¼Ć”±ĄķÄīµÄ×ö·Ø£ŗ ”£
£Ø2£©·Ø¹ś»Æѧ¼ŅPaulSabatier²ÉÓĆ”°×éŗĻ×Ŗ»Æ”±¼¼ŹõŹ¹CO2ŗĶH2ŌŚ“߻ƼĮ×÷ÓĆĻĀÉś³ÉCH4ŗĶH2O”£ŹŌŠ“³öøĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ ”£
£Ø3£©½üĄ“ÓŠæĘѧ¼ŅĢį³öĄūÓĆ”°Ģ¼²¶×½¼¼Źõ”±Ą“½µµĶ¹¤ŅµÉś²śÖŠ¶žŃõ»ÆĢ¼µÄÅÅ·ÅĮ攣”°Ģ¼²¶×½¼¼Źõ”±ŹĒÖøĶعżŅ»¶ØµÄ·½·Ø£¬½«¹¤ŅµÉś²śÖŠ²śÉśµÄCO2·ÖĄė³öĄ“½ųŠŠ“¢“ęŗĶĄūÓĆ”£³£ĄūÓĆ×ćĮæµÄNaOHČÜŅŗĄ“”°²¶×½”±CO2£¬¹ż³ĢČēĻĀĶ¼ĖłŹ¾£Ø²æ·ÖĢõ¼ž¼°ĪļÖŹĪ“±ź³ö£©”£
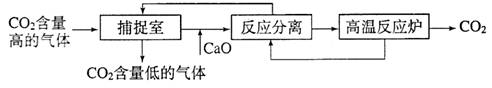
¢Ł²¶×½ŹŅÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ£ŗ ”£
¢Ś°ŃCaO·ÅČė·“Ó¦·ÖĄėŹŅÖŠÓėH2O·“Ó¦£¬ĒėŠ“³öøĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½£ŗ £»ĄūÓĆ“Ė·“Ó¦£¬Ńõ»ÆøĘæÉÓĆ×÷Ź³Ę· ¼Į”£
¢Ū”°·“Ó¦·ÖĄė”±ÖŠ£¬µĆµ½¹ĢĢåĪļÖŹµÄ»ł±¾²Ł×÷ŹĒ £¬øĆ¹ĢĢåŹĒĢ¼ĖįøĘ”£
¢ÜÕūøö¹ż³ĢÖŠ£¬æÉŅŌŃ»·ĄūÓƵÄĪļÖŹÓŠ ”£
£Ø4£©Č”10gĢ¼ĖįøĘ¹ĢĢåøßĪĀ¼ÓČČ£¬Ņ»¶ĪŹ±¼äŗóĶ£Ö¹¼ÓČČ£¬²āµĆŹ£Óą¹ĢĢåÖŠøĘŌŖĖŲµÄÖŹĮæ·ÖŹżĪŖ50%£¬ŌņĻĀĮŠÅŠ¶ĻÕżČ·µÄŹĒ
A.Éś³ÉĮĖ2g¶žŃõ»ÆĢ¼
B£®Ź£Óą¹ĢĢåÖŹĮæĪŖ5g
C£®Éś³ÉĮĖ5.6gŃõ»ÆøĘ
D£®Ź£ÓąĢ¼ĖįøʵÄÖŹĮæĪŖ8g
(1)ĪĀŹŅŠ§Ó¦ ¹āŗĻ×÷ÓĆ ĖęŹÖ¹ŲµĘ£¬·ĻĪļĄūÓĆ(ŗĻĄķ¼“æÉ)?
(2)CO2+4H2 CH4+2H2O?
CH4+2H2O?
(3)¢ŁCO2+2NaOH ===Na2CO3+H2O?¢ŚCaO+H2O ===Ca(OH) 2 øÉŌļ?
¢Ū¹żĀĖ?¢ÜCaO”¢NaOH? (4)A
”¾½āĪö”æ£Ø1£©¶žŃõ»ÆĢ¼ŹĒĪĀŹŅĘųĢ壬æÉŅŌŅżĘšĪĀŹŅŠ§Ó¦£»Ö÷ŅŖĻūŗĶžŃõ»ÆĢ¼ĘųĢåŹĒÖ²ĪļµÄ¹āŗĻ×÷ÓĆ£»ČÕ³£Éś»īÖŠ·ūŗĻ”°µĶĢ¼¾¼Ć”±ĄķÄīµÄ×ö·ØČē£ŗ½ŚŌ¼ÓĆĖ®”¢ĖęŹÖ¹ŲµĘ”¢·ĻĪļĄūÓĆ”¢ÉŁæŖĖ½¼Ņ³µ”¢¶ą×ö¹«¹²Ęū³µµČ”£
£Ø2£©ÓÉĢāøÉĖłøųŠÅĻ¢æÉÖŖ£¬CO2ŗĶH2ŌŚ“߻ƼĮ×÷ÓĆĻĀÉś³ÉCH4ŗĶH2O£¬·“Ó¦±ķ“ļŹ½ĪŖCO2+4H2 CH4+2H2O?”£
CH4+2H2O?ӣ
£Ø3£©¢Ł²¶×½ŹŅŹĒĄūÓĆNaOHÓėCO2·“Ó¦Ą“”°²¶×½”±CO2ĘųĢ壬CO2+2NaOH ===Na2CO3+H2O”£
¢ŚCaOÓėĖ®·“Ó¦µÄ·½³ĢŹ½CaO+H2O ===Ca(OH) 2 £¬æÉĄūÓŚŃõ»ÆøĘÓėĖ®·“Ó¦£¬æɽ«Ńõ»ÆøĘ×÷Ź³Ę·µÄøÉŌļ¼ĮŹ¹ÓĆ”£
¢Ū”°·“Ó¦·ÖĄė”±ÖŠ£¬ŹĒ½«²»ČÜŠŌ¹ĢĢåÓėŅŗĢå·ÖĄė£¬¹ŹŹ¹ÓƵĻł±¾²Ł×÷ŹĒ¹żĀĖ”£
¢ÜÓÉĶ¼Ź¾æÉŅŌ擳ö£¬æÉŅŌŃ»·Ź¹ÓƵďĒCaO”¢NaOH”£
(4)øł¾ŻÖŹĮæŹŲŗć¶ØĀÉæÉÖŖ£¬·“Ó¦Ē°ŗóŌŖĖŲµÄÖŹĮæ²»±ä£¬·“Ó¦Ē°øĘŌŖĖŲµÄÖŹĮæ=10g”Į =4g
=4g
¹Ź·“Ó¦ŗóŹ£Óą¹ĢĢåÖŹĮæĪŖ4g”Ā50%=8g£¬ÓÉ»Æѧ·½³ĢŹ½CaCO3  CaO+CO2”üæÉÖŖ¹ĢĢå¼õÉŁµÄÖŹĮæĪŖĘųĢ嶞Ńõ»ÆĢ¼µÄÖŹĮæ£Ø10g-8g£©=2g”£Éś³ÉCaOµÄÖŹĮæĪŖ2g/44”Į56”Ł5.6g£¬¹ŹÖ»ÓŠAÕżČ·”£
CaO+CO2”üæÉÖŖ¹ĢĢå¼õÉŁµÄÖŹĮæĪŖĘųĢ嶞Ńõ»ÆĢ¼µÄÖŹĮæ£Ø10g-8g£©=2g”£Éś³ÉCaOµÄÖŹĮæĪŖ2g/44”Į56”Ł5.6g£¬¹ŹÖ»ÓŠAÕżČ·”£



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
| ||
| ||

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
| ||
| ||
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ½õÖŻ ĢāŠĶ£ŗĪŹ“šĢā
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com