����������ɫ�д̼�����ζ���壩�������������������������Ҫ�Ĵ�����Ⱦ��ҹ�����ʮ�����ӻ��������������������̨ÿ�조������������Ҫ����ȫ��42����Ҫ���п��������ձ������������ǹ�ע�Լ����������Ŀǰ���йز��Ÿ���SO
2��Br
2���壩��H
2O�Ķ�����Ӧ���ⶨ������SO
2�ĺ�����������һԭ����ͨ���ڵ����з�����Ӧʱ�����ı仯������ȷ�ⶨ����������������ĺ�����ijУ��ѧ����С��Ϊ�ⶨУ����������SO
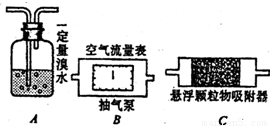
2������������ĺ���������������Ӧԭ�����������װ�ã�
��1��������������ȷ�ⶨ�������õĿ����������ⶨ���������P����������ʱ����ͼ��ʾ����װ�����ӵ�˳���ǣ�����ţ�______��
��2��Ϊȷ�ⶨSO
2�ĺ�����ʵ��ʱ���۲쵽______ʱ��Ӧ�����رճ����ã���Ӧԭ����
Br
2+2H
2O+SO
2=H
2SO
4+2HBr
����ɫ�� ����ɫ��
��3����Ҫ�ⶨ����������������ĺ�������Ҫ���ʵ��ʱ���������⣬��Ҫ��______������ţ���
a��ʵ��ǰB������ b��ʵ���B ������
c��ʵ��ǰC ������ d..ʵ���C ��������




 ������ҵ��ͬ����ϰ��ϵ�д�
������ҵ��ͬ����ϰ��ϵ�д�