
| �������� | ʵ������ | ���ۼ���ѧ����ʽ |
| ȡ������ʢ�ж�����̼�ļ���ƿ�� | ______ | ��������______��Һ�������ƣ� ��صĻ�ѧ����ʽ______ |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| �������� | ʵ������ | ���ۼ���ѧ����ʽ |
| ȡ�����Թ��У� �μ�ϡ���� �μ�ϡ���� |
�����ݲ��� �����ݲ��� |
�������ȷ����Ӧ�Ļ�ѧ����ʽΪ�� Na2CO3+2HCl�T2NaCl+H2O+CO2�� �������ȷ����Ӧ�Ļ�ѧ����ʽΪ�� Na2CO3+2HCl�T2NaCl+H2O+CO2�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| �������� | ʵ������ | ���ۼ���ѧ����ʽ |
| ȡ������ʢ�ж�����̼�ļ���ƿ�� | �а�ɫ���� �а�ɫ���� |
������ȷ ��صĻ�ѧ����ʽ CO2+Ba��OH��2�TBaCO3��+H2O CO2+Ba��OH��2�TBaCO3��+H2O |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| �������� | ʵ������ | ���ۼ���ѧ����ʽ |
| ȡ������ʢ�ж�����̼�ļ���ƿ�� | �Լ������ �Լ������ |
�������� �������� �������� ��Һ�������ƣ���صĻ�ѧ����ʽ CO2+Ba��OH��2�TBaCO3��+H2O CO2+Ba��OH��2�TBaCO3��+H2O |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
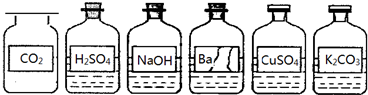
ij��ȤС��ͬѧ����ʵ��̨������ڷŵ�ҩƷ�У�����ͼ������һ�Լ�ƿ�ı�ǩ����

��������⡿��ƿ�Լ��ijɷ���ʲô��
����Ʋ�ʵ�顿
��1��С���ýྻ����IJ�����պȡ����Һ�ε�ʪ���pH��ֽ�ϣ����pH=10�����ж�����һ�ּ�����Һ�������IJ���������������������۵����ۣ���ȷ���� ��
A��������ȷ�������ȷ���ҽ�����ȷ B����������ȷ�����ƫС����������ȷ
C����������ȷ�����ƫ���۲���ȷ D��������ȷ�������ȷ�������۲���ȷ
��2��С����Ϊ����Ba(OH)2��Һ��С����Ϊ����Ba(NO3)2��Һ��Ҫ��һ��ȷ�����ijɷ֣�С����Ϊֻ��ѡ����ͼ���е��Լ����ܰ�����������������ֽ�����ʵ�顣
| �������� | ʵ������ | ���ۼ���ѧ����ʽ |
| ȡ������ʢ�ж�����̼�ļ���ƿ�� |
| ������ȷ ��صĻ�ѧ����ʽ |
��֪ʶ������
��3������д�����з������������Ļ�ѧ����ʽ��
���а�ɫ�������кͷ�Ӧ��
������ͭ��Һ������������Һ֮�䷴Ӧ��
��4���ҷ���NaOH��H2SO4��Һ�ܷ������ֽⷴӦ����Ϊ��Һ�е�H+��OH—���ܴ������棻�һ���������Һ�� �� �� �� �������ӷ��ţ�Ҳ���ܴ������档
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij��ȤС��ͬѧ����ʵ��̨������ڷŵ�ҩƷ�У�����ͼ������һ�Լ�ƿ�ı�ǩ����

��������⡿��ƿ�Լ��ijɷ���ʲô��
����Ʋ�ʵ�顿
��1��С���ýྻ����IJ�����պȡ����Һ�ε�ʪ���pH��ֽ�ϣ����pH=10�����ж�����һ�ּ�����Һ�������IJ���������������������۵����ۣ���ȷ���� ��
A��������ȷ�������ȷ���ҽ�����ȷ B����������ȷ�����ƫС����������ȷ
C����������ȷ�����ƫ���۲���ȷ D��������ȷ�������ȷ�������۲���ȷ
��2��С����Ϊ����Ba(OH)2��Һ��С����Ϊ����Ba(NO3)2��Һ��Ҫ��һ��ȷ�����ijɷ֣�С����Ϊֻ��ѡ����ͼ���е��Լ����ܰ�����������������ֽ�����ʵ�顣
| �������� | ʵ������ | ���ۼ���ѧ����ʽ |
| ȡ������ʢ�ж�����̼�ļ���ƿ�� |
| ������ȷ ��صĻ�ѧ����ʽ |
��֪ʶ������
��3������д�����з������������Ļ�ѧ����ʽ��
���а�ɫ�������кͷ�Ӧ��
������ͭ��Һ������������Һ֮�䷴Ӧ��
��4���ҷ���NaOH��H2SO4��Һ�ܷ������ֽⷴӦ����Ϊ��Һ�е�H+��OH—���ܴ������棻�һ���������Һ�� �� �� �� �������ӷ��ţ�Ҳ���ܴ������档
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com