
��ij���������֪�û�����п��ܺ���NaOH��Mg(OH)2��CaCO3��Cu(NO3)2��CuSO4���������е����ֻ���֡�����ͼ��ʾ����ʵ�飬���ֵ�������ͼ����������������з����ķ�Ӧ��ǡ����ȫ��Ӧ����
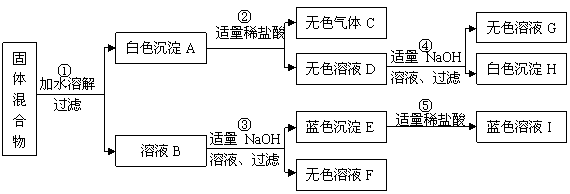 |
����ʵ����̺ͷ�����������д���¿հף�
��1������ɫʯ����Һ��������C����Һ��__________ ɫ��
��2��ʵ��֤��������ҺB�У�������������������(д���ӷ���)_____________��
��3��ʵ����̢��з�����Ӧ�Ļ�ѧ����ʽΪ_________________________________��
��4������ʵ�������֤������������϶��������������� ��
��5������������������������У�������ȷ�������������ǣ�д��ѧʽ��______________��Ҫ��һ�������ù����������ɣ�������ҺF��ʵ�飬���Ҫ˵��ʵ��������衢������������__________________ _____
_________________________________________________________________________ ��
 �ο�����������100��ϵ�д�
�ο�����������100��ϵ�д� �Űٷֿ�ʱ����ϵ�д�
�Űٷֿ�ʱ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
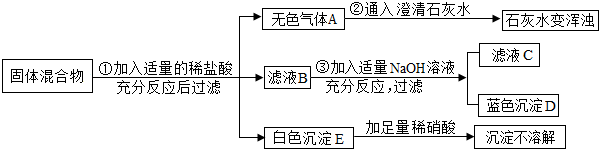
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
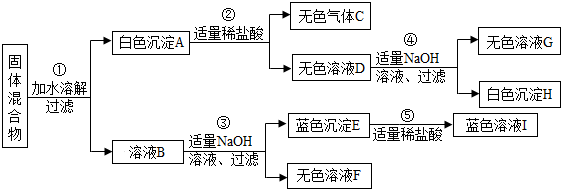
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
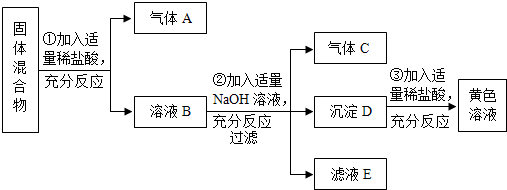
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com