�ᡢ�����������Ҫ�Ļ�����ռ����������Ƶ��׳ƣ�����һ����Ҫ�Ļ���ԭ�ϣ��㷺Ӧ���ڷ�������ֽ�ȹ�ҵ��
��1���ռ��ڹ�ҵ��ͨ���õ�ⱥ��ʳ��ˮ�ķ�����ȡ��������ռ��⣬����H
2��Cl
2����д����ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ
2NaCl+2H
2O
2NaOH+Cl
2��+H
2��
2NaCl+2H
2O
2NaOH+Cl
2��+H
2��
��
��2���ռ��ڿ����з��û���ʣ��������һ��ʵ�飬���ܼ����ռ���ʵ�ͬʱ�����ܳ�ȥ���ʣ��û�ѧ����ʽ��ʾ
Na2CO3+Ca��OH��2=CaCO3��+2NaOH
Na2CO3+Ca��OH��2=CaCO3��+2NaOH
��
��3��������кܶͬ���ص㣬������ᶼ�ܹ������кͷ�Ӧ�������ᶼ��ʹ���ָʾ����ɫ�ȣ�������д����ͼ�����ɺ����ʷ���Ĺ�ͬ�㣨��дһ����
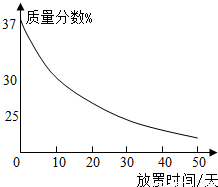
��4����ͬѧ�ù����ռ��ˮ����100g 18.5%��NaOH��Һ�����������������ռ���Һʱ�IJ�����
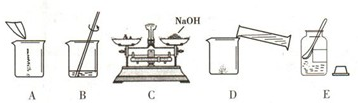
����ͼ��ʾ����ű�ʾ��ȷ���Ƹ���Һ�IJ���˳��Ϊ
ECADB
ECADB
��
����ͼC�����������Ϊ15g������Ķ���Ϊ3.5g����С���Ƶõ�������������ʵ��Ϊ
11.5
11.5
g��
��5��ͬѧ����pH��ֽ�ⶨ�����Ƶ�����������Һ�����ȣ���ȷ�ķ�����
�ò������������Ƶ�����������Һ����pH��ֽ�ϣ���ͣ��ͱ���ɫ�Ƚϣ�������Һ��pH
�ò������������Ƶ�����������Һ����pH��ֽ�ϣ���ͣ��ͱ���ɫ�Ƚϣ�������Һ��pH
��
��6����8g��������Ϊ20%������������Һ��22gij����ǡ����ȫ�кͣ��Լ��㷴Ӧ��������Һ���ʵ�����������




 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д�