
،¾جâؤ؟،؟»¯ر§ہدت¦½»¸ّؤ³ر§ذ£رذ¾؟ذشر§د°ذ،×éز»¸ِبخخٌ£؛²â¶¨تµرéتزہïز»ئ؟¾أضأµؤ![]() ¹ججهتا·ٌ±نضت£®¸أذ،×éµؤح¬ر§½ّذذءثبçدآتµرéج½¾؟£؛
¹ججهتا·ٌ±نضت£®¸أذ،×éµؤح¬ر§½ّذذءثبçدآتµرéج½¾؟£؛
جل³ِختجâ£؛²آدë![]() £؛أ»±نضت£¬ب«²؟تا
£؛أ»±نضت£¬ب«²؟تا![]() £»
£»
²آدë![]() £؛²؟·ض±نضت£¬________£»
£؛²؟·ض±نضت£¬________£»
²آدë![]() £؛حêب«±نضت£¬ب«²؟تا
£؛حêب«±نضت£¬ب«²؟تا![]() £®
£®
²éشؤ×تءد£؛²؟·ضبـز؛شع³£خآدآµؤ![]() بçدآ£؛
بçدآ£؛
رخبـز؛ |
|
|
|
| µبسع | ´َسع | µبسع |
ةè¼ئ·½°¸²¢½ّذذتµرé£؛اëؤمسëثûأاز»ئً¹²ح¬حê³ة£¬²¢»ط´ًثù¸ّختج⣮
تµرé²½ضè | تµرéدضدَ | تµرé½لآغ |
¢ظ³ئب،ةدتِاâرُ»¯ؤئ¹ججهرùئ· | ²ْةْ°×ة«³ءµي | ثµأ÷¾أضأ¹ججهضذ£¬ز»¶¨؛¬سذ ________£¨جر§ت½£©£® |
¢عسأ²£ء§°ôص؛ب،ةظء؟¢ظضذ³ن·ض·´س¦؛َµؤةد²ماهز؛µخشعز»ذ،؟é |
| ثµأ÷¾أضأ¹ججهضذ£¬»¹ز»¶¨؛¬سذ ________ £¨جر§ت½£©£® |
ةدتِ²½ضè¢ظضذ£¬³ئء؟اâرُ»¯ؤئ¹ججهرùئ·سأµ½µؤ²£ء§زائ÷أû³ئتا________£¬µخ¼س¹ء؟![]() بـز؛µؤؤ؟µؤتا________
بـز؛µؤؤ؟µؤتا________
تµرé½لآغ£؛ح¨¹تµر飬ثµأ÷ةدتِ²آدëضذ________تاصب·µؤ£®
حطص¹£؛¸أذ،×éح¬ر§خھ²â¶¨³ِ¸أ![]() ¹ججهµؤ±نضت³ج¶ب£¬¼جذّ½«ةدتِ°×ة«³ءµي¹آث،¢د´¾»،¢¸ةش³ئµأئنضتء؟خھ
¹ججهµؤ±نضت³ج¶ب£¬¼جذّ½«ةدتِ°×ة«³ءµي¹آث،¢د´¾»،¢¸ةش³ئµأئنضتء؟خھ![]() £¬شٍشرùئ·ضذ
£¬شٍشرùئ·ضذ![]() µؤضتء؟·ضتخھ________ £¨±£ءôز»خ»ذ،ت£©£®
µؤضتء؟·ضتخھ________ £¨±£ءôز»خ»ذ،ت£©£®
·´ث¼£؛¾أضأµؤاâرُ»¯ؤئ±نضتµؤشزٍتا________£¨سأ»¯ر§·½³جت½±يت¾£©£®
،¾´ً°¸،؟![]() ؛ح
؛ح![]() £®
£®![]()
![]() ةص±بأج¼ثلؤئب«²؟²خ¼س·´س¦²آدë
ةص±بأج¼ثلؤئب«²؟²خ¼س·´س¦²آدë![]()
![]()
![]()
،¾½âخِ،؟
اâرُ»¯ؤئؤـخüتص؟صئّضذµؤ¶رُ»¯ج¼±نضت£¬؟ةؤـ²؟·ض±نضت£¬ز²؟ةؤـب«²؟±نضت،£شع¼ىرéاâرُ»¯ؤئتا·ٌ±نضتت±£¬µخ¼سرخثلسذئّإف²ْةْ،£بôزھض¤أ÷تا²؟·ض±نضت£¬شٍدب¼ىرéسذج¼ثلؤئ£¬؟ةزش¼سبë¹ء؟آب»¯±µبـز؛£¬ح¨¹²ْةْ°×ة«³ءµيہ´ض¤أ÷£¬ح¨¹¼سبë¹ء؟آب»¯±µبـز؛شع³ب¥ج¼ثلؤئض®؛َ¼ىرéبـز؛دش¼îذش£¬ثµأ÷»¹سذاâرُ»¯ؤئ£»¸ù¾فةْ³ة³ءµيµؤضتء؟½ل؛د·´س¦µؤ»¯ر§·½³جت½¼ئثم½â´ً£»¸ù¾فاâرُ»¯ؤئµؤذشضتہ´½ّذذ½â´ً،£
²آدë¢ٍ£؛²؟·ض±نضت£¬¼ب؛¬سذNaOHسض؛¬سذNa2CO3£»تµرé½لآغ¢ظدٍبـز؛ضذµخ¼سآب»¯±µبـز؛ضء¹ء؟£¬³ن·ض·´س¦؛َ£¬¾²ضأ،£²ْةْ°×ة«³ءµي£¬ثµأ÷¾أضأ¹ججهضذ£¬ز»¶¨؛¬سذNa2CO3£»¢عةْ³ة³ءµي؛َت£سàبـز؛µؤpH=11£¬بـز؛دش¼îذش£¬ثµأ÷¾أضأ¹ججهضذ£¬»¹ز»¶¨؛¬سذNaOH£»اâرُ»¯ؤئ¹ججهز׳±½â£¬سذا؟ءزµؤ¸¯ت´ذش£¬¹تس¦·إشعةص±ضذ³ئء؟£»µخ¼س¹ء؟BaCl2بـز؛µؤؤ؟µؤتابأج¼ثلؤئب«²؟²خ¼س·´س¦£»تµرé½لآغ£؛ح¨¹تµرé؟ةضھ£¬ش¹ججهضذ¼ب؛¬سذNaOHسض؛¬سذNa2CO3£¬ثµأ÷ةدتِ²آدëضذ²آدë¢ٍتاصب·µؤ£»
حطص¹£؛ةèرùئ·ضذج¼ثلؤئµؤضتء؟خھx،£
Na2CO3+BaCl2=BaCO3،+2NaCl
106 197
x 4.0g
![]()
x=2.15g
رùئ·ضذاâرُ»¯ؤئµؤضتء؟خھ10g-2.15g=7.85g£¬شٍشرùئ·ضذNaOHµؤضتء؟·ضتخھ![]() =78.5£¥£»·´ث¼£؛¾أضأµؤاâرُ»¯ؤئ±نضتµؤشزٍتا2NaOH+CO2=Na2CO3+H2O،£
=78.5£¥£»·´ث¼£؛¾أضأµؤاâرُ»¯ؤئ±نضتµؤشزٍتا2NaOH+CO2=Na2CO3+H2O،£


 أûذ£؟خجأدµءذ´ً°¸
أûذ£؟خجأدµءذ´ً°¸
| ؤ꼶 | ¸كضذ؟خ³ج | ؤ꼶 | ³ُضذ؟خ³ج |
| ¸كز» | ¸كز»أâ·ر؟خ³جحئ¼ِ£، | ³ُز» | ³ُز»أâ·ر؟خ³جحئ¼ِ£، |
| ¸ك¶ | ¸ك¶أâ·ر؟خ³جحئ¼ِ£، | ³ُ¶ | ³ُ¶أâ·ر؟خ³جحئ¼ِ£، |
| ¸كب | ¸كبأâ·ر؟خ³جحئ¼ِ£، | ³ُب | ³ُبأâ·ر؟خ³جحئ¼ِ£، |
؟ئؤ؟£؛³ُضذ»¯ر§ ہ´ش´£؛ جâذح£؛
،¾جâؤ؟،؟دآءذختجâµؤرذ¾؟ضذ£¬خ´ہûسأ¶ش±بتµرéث¼دë·½·¨µؤتا(،،،،)
A.  رذ¾؟؟صئّضذرُئّ؛¬ء؟
رذ¾؟؟صئّضذرُئّ؛¬ء؟
B.  رذ¾؟·ض×سµؤشث¶¯
رذ¾؟·ض×سµؤشث¶¯
C. 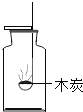 رذ¾؟ؤ¾ج؟شعرُئّضذب¼ةص
رذ¾؟ؤ¾ج؟شعرُئّضذب¼ةص
D.  رذ¾؟¶رُ»¯ج¼سëث®µؤ·´س¦
رذ¾؟¶رُ»¯ج¼سëث®µؤ·´س¦
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛³ُضذ»¯ر§ ہ´ش´£؛ جâذح£؛
،¾جâؤ؟،؟دآح¼ضذ£¬¢ظتاؤئشھثطشعشھثطضـئع±يضذµؤذإد¢£¬A،¢B،¢Cتابضضء£×سµؤ½ل¹¹ت¾زâح¼،£
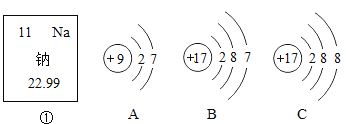
تش»ط´ًدآءذختî}£؛
£¨1£©ؤئشھثطµؤش×سذٍتخھ £»
£¨2£©A،¢B،¢Cضذتôسعح¬ضضشھثطµؤء£×ستا £¨ذ´×ضؤ¸±à؛إ£©£»
£¨3£©A؛حBء½ضضء£×سµؤ دàح¬£¬ثùزشثüأاسذدàثئµؤ»¯ر§ذشضت£»
شع»¯ر§·´س¦ضذثüأابفز× £¨جî،°µأµ½،±»ٍ،°ت§ب¥،±£©µç×س£»
£¨4£©Cثù±يت¾µؤء£×ستا £¨جî،°ش×س،±»ٍ،°ہë×س،±£©،£
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛³ُضذ»¯ر§ ہ´ش´£؛ جâذح£؛
،¾جâؤ؟،؟دٍ160gءٍثلحبـز؛ضذ£¬ضًµخ¼سبë40g NaOHبـز؛£¬ا،؛أحêب«·´س¦£¬ةْ³ة4.9g³ءµي£¬»ط´ًدآءذختجâ£؛

£¨1£©ا،؛أحêب«·´س¦؛َ£¬بـز؛µؤضتء؟خھ g،£
£¨2£©½«·´س¦½لتّ؛َµؤ»ى؛دخï¹آث£¬آثز؛ضذبـضتتا £¨جر§ت½£©،£
£¨3£©اَشءٍثلحبـز؛ضذبـضتµؤضتء؟·ضت£¨ذ´³ِ¼ئثم¹³ج£©،£
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛³ُضذ»¯ر§ ہ´ش´£؛ جâذح£؛
،¾جâؤ؟،؟ض¸³ِدآءذ²ظ×÷؟ةؤـµ¼ضآµؤ²»ء¼؛َ¹û£®
![]() ؟é×´¹ججه´ستش¹ـ؟عتْض±ح¶بëتش¹ـؤع________£®
؟é×´¹ججه´ستش¹ـ؟عتْض±ح¶بëتش¹ـؤع________£®
![]() دٍب¼×إµؤ¾ئ¾«µئہïجي¼س¾ئ¾«________£®
دٍب¼×إµؤ¾ئ¾«µئہïجي¼س¾ئ¾«________£®
![]() ب،ز؛؛َµؤµخ¹ـئ½·إ»ٍµ¹ضأ________£®
ب،ز؛؛َµؤµخ¹ـئ½·إ»ٍµ¹ضأ________£®
![]() ±»¼سببµؤ²£ء§بفئ÷حâ±عسذث®________£®
±»¼سببµؤ²£ء§بفئ÷حâ±عسذث®________£®
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛³ُضذ»¯ر§ ہ´ش´£؛ جâذح£؛
،¾جâؤ؟،؟ز½ش؛¾³£سأز»ضض°µ×دة«دû¶¾·غ¶ش»·¾³½ّذذدû¶¾،£ب،ةظء؟¸أدû¶¾·غ½ّذذتµر飬تµرé¹³جبçح¼ثùت¾£¬ئنضذئّجهBؤـت¹´ّ»ًذاµؤؤ¾جُ¸´ب¼،£

اë»ط´ًدآءذختجâ£؛
(1)ذ´³ِدآءذخïضتµؤ»¯ر§ت½£؛A___£»B___£»C___،£
(2)ذ´³ِدآءذ»¯ر§·½³جت½£؛
؛عة«¹ججهCسëخقة«ز؛جهµؤ·´س¦£؛_______________£»
؛ىء×شعخقة«ئّجهBضذب¼ةصµؤ·´س¦£؛_______________،£
(3)ةدتِ×ھ»¯¹³جضذ£¬°×ة«¹ججهخïضتµؤأû³ئتا_________،£
(4)Aتـبب·ض½â؛َµأµ½µؤ¹ججهخïضتضذ£¬ؤـ¹»بـسعث®µؤخïضتµؤ»¯ر§ت½خھ_________،£
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛³ُضذ»¯ر§ ہ´ش´£؛ جâذح£؛
،¾جâؤ؟،؟دآ±يءذ³ِءثدُثل¼طشع²»ح¬خآ¶بت±µؤبـ½â¶ب£®
خآ¶ب/،و | 0 | 10 | 20 | 30 | 40 | 50 | 60 |
بـ½â¶ب/g | 13.3 | 20.9 | 31.6 | 45.8 | 63.9 | 85.5 | 110 |
ؤ³ذثب¤ذ،×é×ِءثزشدآتµرé£؛

£¨1£©ةدتِبـز؛ضذµؤبـ¼ءتا________£¬ةدتِتµرé¹³جضذµأµ½µؤبـز؛ز»¶¨تôسع²»±¥؛حبـز؛µؤتا________£¨جîت×ضذٍ؛إ£©£¬¢فثù¶شس¦µؤتµرéدضدَتا________£®
£¨2£©تµرé¹³جضذ£¬بـضتµؤضتء؟·ضتز»¶¨دàµبµؤتا________£¨جîت×ضذٍ؛إ£©£®
£¨3£©خھءث·ہض¹ث®جهخغب¾£¬ذ،×éح¬ر§جل³ِءثبçدآ·½°¸´¦ہيتµرé؛َµؤ·دز؛£؛ ²½ضèI£؛½«¢ف¼جذّہنب´ضء0،و£¬¹آث£¬¹²؟ة»طتصµأµ½KNO3¹ججه________g£®
²½ضèII£؛دٍآثز؛ضذ¼سبë________gث®د،تح£¬µأµ½بـضتµؤضتء؟·ضتخھ1%µؤKNO3د،بـز؛£¬½«´ثبـز؛×ِخھ»¯·تت¹سأ£®
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛³ُضذ»¯ر§ ہ´ش´£؛ جâذح£؛
،¾جâؤ؟،؟ذ،؟ئح¬ر§¶ش¹م؟عئ؟²ْةْءثذثب¤£¬شع²»ح¬µؤتµرéضذسذ×إ²»ح¬µؤ×÷سأ£¬زشدآ½âتحصب·µؤتا
تµرé ×°ضأ |
|
|
|
|
½âتح | سأسعإإ؟صئّ·¨تص¼¯اâئّµؤ×°ضأ | سأسع¸ةشï¶رُ»¯ج¼µؤ×°ضأ | سأسع²âء؟رُئّجه»µؤ×°ضأ | سأسعز½ش؛¸ّ²،بث¹©رُئّµؤ×°ضأ |
ر،دî |
|
|
|
|
A. ![]() B.
B. ![]() C.
C. ![]() D.
D. ![]()
²é؟´´ً°¸؛ح½âخِ>>
؟ئؤ؟£؛³ُضذ»¯ر§ ہ´ش´£؛ جâذح£؛
،¾جâؤ؟،؟اëبدصو»طزنخïضتسëئن¹¹³ةء£×سض®¼نµؤ¹طدµ£¬»ط´ًدآءذختجâ£؛
£¨1£©½ًتôجْتاسة ¹¹³ةµؤ£¬آب»¯ؤئتاسة ¹¹³ةµؤ£¬¶رُ»¯ج¼تاسة ¹¹³ةµؤ،££¨جî ،°ش×س،±،¢،°·ض×س،±»ٍ ،°ہë×س،±£©
£¨2£©ش×سµأµ½»ٍت§ب¥µç×س؛َذخ³ةہë×س،£ؤ³ء£×سµؤ½ل¹¹ت¾زâح¼خھ
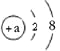
¢ظµ±a= ت±£¬¸أء£×ستاش×س£¬¸أش×سخ»سعشھثطضـئع±يضذµع ضـئع،£
¢عµ±a=8ت±£¬¸أء£×ستا £¨جî،°ش×س،±،¢،°رôہë×س،±»ٍ ،°زُہë×س،±£©،£
²é؟´´ً°¸؛ح½âخِ>>
°ظ¶بضآذإ - ء·د°²لءذ±ي - تشجâءذ±ي
؛±±ت،»¥ءھحّخ¥·¨؛ح²»ء¼ذإد¢¾ظ±¨ئ½ج¨ | حّةدسذ؛¦ذإد¢¾ظ±¨×¨اّ | µçذإص©ئ¾ظ±¨×¨اّ | ةوہْت·ذéخقض÷زهسذ؛¦ذإد¢¾ظ±¨×¨اّ | ةوئَاضب¨¾ظ±¨×¨اّ
خ¥·¨؛ح²»ء¼ذإد¢¾ظ±¨µç»°£؛027-86699610 ¾ظ±¨ستدن£؛58377363@163.com