
 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
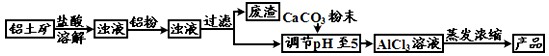
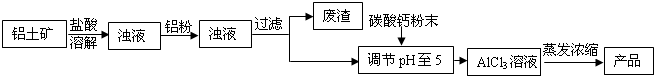
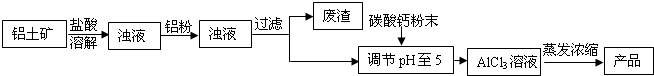
��ʽ�Ȼ�����ij�ָ�Ч��ˮ������Ҫ�ɷ֡�ʵ��������������(��Ҫ��Al2O3����������Fe2O3��������������)��ȡ��ʽ�Ȼ����Ĺ������£�
![]()
(1)�ܽ�����У���Ҫʹ����������Ϊ15%�����ᣬ����������������Ҫ______mL��30%��Ũ����(�ܶ�ԼΪ1.15g/cm3)��115g����ˮ��
(2)�����ܽ�����������У�������Ӧ�Ļ�ѧ����ʽΪ________________________��
(3)���������۵���Ҫ�����ǣ���ȥ��Һ�е�______��
(4)��CaCO3��ĩ�������ǵ���pH��5����д����pH��ֽ�ⶨ��ҺpH�IJ������ڰ״ɰ����Ƭ�Ϸ�һСƬpH��ֽ��____________________________________���ó�����Һ��pH��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com