
ʵ����һƿ�¹�����Ũ���ᣬ���ǩ�IJ���������ͼ��ʾ��

��1������ƿ���ỹû�п���ʹ�ã�ƿ����Һ������Ϊ________��g��
��2����Ҫ����9.8%��ϡ����400g�������ø�Ũ����________mL��������������0.1����
��3������ϡ����Ĺ����У������õ���Ͳ����ͷ�ιܡ��ձ��⣬����Ҫ�õ�������������________ ��
��4������ȡŨ����ʱ�������ӵķ����������Ƶ���Һ������������________9.8%�����������=����������
��5����ȡ��9.8%��ϡ����100g��������þ��ַ�Ӧ��������Һ������Ϊ________g��
92021.7��������102.2 �������� ��1������ƿ���ỹû�п���ʹ�ã���Ũ��������տ����е�ˮ�֣���ƿ����Һ������Ϊ500mL��1.84g/mL=920g�� ��2����Һϡ�����������������䣬����Ҫ98%��Ũ���������Ϊx������98%��x=400g��9.8%�����x=40g����ҪŨ������������21.7mL�� ��3����Ũ���������ձ�������ע��ˮ�У���ų��������ȣ�Ϊ...
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ���ʰ��滯ѧ���꼶�ϲ�4.3��ʯȼ�ϵ����� ��Ԫ����� ���ͣ���ѡ��
�����ҹ��ۺ�ʵ������ǿ����������ˮƽ��������ߣ��������߽�ǧ��������˵������ȷ���ǣ�������
A. ������кǼ����ɺϽ��Ƴɣ���̥���ɺϳ����Ƴ�
B. ��������ѹ����Ȼ����Һ��ʯ������ȼ�ϣ����Լ��ٶԿ�������Ⱦ
C. �ٿ�˽�ҳ��������������ͨ���ߣ����ϡ����ܼ�̼���Ļ�������
D. �ڼ���վ�������ŵ����͵���ζ������Ϊ���Ӽ����һ���ľ���
D �������� ������������кǼܶ����ɺϽ���ɵģ���̥�Ǻϳ����Ƴɵģ�����A��ȷ�����ͻ����ȼ�ջ��ͷų������������������к����壬����Ȼ����Һ��ʯ����ȼ�գ��������ʲ����Ľ��٣������ڼ����������Ⱦ������B��ȷ���ٿ�˽�ҳ��������������ͨ���ߣ���������Դ�����ģ���������Ⱦ����ŷţ����ϡ����ܼ�̼���Ļ���������C��ȷ���ڼ���վ�������ŵ����͵���ζ������Ϊ�����ڲ��ϵ��˶�������D����...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ��ʯ��2018����꼶��ѧ�����п��Ի�ѧ�Ծ� ���ͣ������
�����벻��ˮ������ˮ��֪ʶ���ճ����������Ź㷺��Ӧ�á�
��1���豭�ڵ�ɴ�����ɽ���Ҷ���ˮ���룬�������ã���������õ���___________��
��2�����ƾ�ˮ���г�����__________�����ڳ�ȥˮ�е���ζ��ɫ�أ�
��3������ˮ���ö�����������ɱ�����������ȵĻ�ѧʽΪ__________��
��4����ũ�峣�þ�ˮ��������ˮ������___________�ɼ��龮ˮ�Ƿ�ΪӲˮ��
��5�����н�ˮ��ʩ��ȷ����______________��
A��ũҵ�ϸĴ�ˮ����Ϊ�ι� B��������ˮ����
C���ô�����ˮ��ϴ���� D��ʹ�ý�ˮ��ͷ
��6��Ӳˮ������������������㣬�ճ������г���_______������Ӳˮת��Ϊ��ˮ��
���˻���̿ClO2����ˮABD��� �������� (1)�豭�ڵ�ɴ�����ɽ���Ҷ���ˮ���룬�������ã���������õĻ�ѧԭ���ǹ��ˣ� (2)���ƾ�ˮ���г��������̿�����ڳ�ȥˮ�е���ζ��ɫ�أ� (3)д��ѧʽͨ��������Ԫ�ط���д���ұߣ���һ��Ԫ�صķ���д�����ɶ������ȶ�����֪��ѧʽ�и�ԭ�ӵĸ��������Զ������ȵĻ�ѧʽΪClO2�� (4)���÷���ˮ������Ӳˮ����ˮ�� ...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���ʰ�����꼶�ϲữѧ 3.1�������ʵ�����Ԫ����� ���ͣ���ѡ��
�йط��Ӻ�ԭ�ӵ�������ȷ���ǣ�������
A. ���ӿ�ֱ�ӹ������ʣ���ԭ�Ӳ�����
B. �����ǻ�ѧ�仯�е���С��
C. ���ӿ����ٷ֣�ԭ�Ӳ������ٷ�
D. �����DZ������ʻ�ѧ���ʵ�һ����
D �������� ����A����Ӻ�ԭ�Ӷ�����ֱ�ӹ������ʣ�ϡ��������ǵ�ԭ�ӷ��ӣ�A�����B��ԭ���ǻ�ѧ�仯�е���С����B�����C��ԭ���ڻ�ѧ�仯�в����ٷ֣��������仯�пɷ֣�C�����D����ȷ����ѡD.�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���ʰ��滯ѧ���꼶�ϲ�5.2�����غ㶨����ϰ�� ���ͣ�������
ij��ȤС��Ϊ�ⶨһ��ʯ��ʯ��Ʒ��̼��Ƶ���������������ʵ�飺��12�˵Ĺ�����Ʒ�в��ϼ���ϡ���ᣬֱ��û�����ݲ���������ȥϡ����100�ˡ�������������ͼ��ʾ��
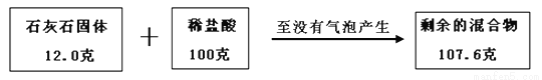
ע���ٸ÷�Ӧ�У���Ʒ�е����ʲ��μӷ�Ӧ��̼�����ȫ��Ӧ���ڼ���������һλС����
��1�����������غ㶨�ɣ���Ӧ���ɶ�����̼������Ϊ________g��
��2���������Ʒ��̼��Ƶ���������________����д��������̣�
4.4g83.3% �������� ������������غ㶨�ɣ���Ӧǰ�����ʵ����������䣬���ɶ�����̼������Ϊ100g+12.0g107.6g=4.4g������Ʒ��̼��Ƶ�����Ϊx�� ��CaCO3+ 2HCl ==CaCl2 + H2O + CO2�� 100 44 x 4.4g 100/x=44/4.4g x=10g ��Ʒ��̼��Ƶĺ���Ϊ10g��12.0g��100%��8...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���ʰ��滯ѧ���꼶�ϲ�5.2�����غ㶨����ϰ�� ���ͣ���ѡ��
ƫ�����£�C2H8N2����N2O4��Ӧ�ų��������ܰѻ������̫�գ��û�ѧ����ʽΪC2H8N2+2N2O4��
2X��+3N2��+4H2O��������˵����ȷ���ǣ� ��
A. X�Ļ�ѧʽΪCO B. ƫ��������̼����������Ϊ40%
C. N2O4�к���1�������� D. ƫ��������̼ԭ�ӡ���ԭ�ӡ���ԭ�ӹ���
B �������� �������غ㶨�ɿ�֪����ѧ��Ӧǰ��ԭ�ӵ����ࡢ�������䣬��Ӧǰ��8����ԭ�ӡ�2��̼ԭ�ӡ�8����ԭ�ӡ�6����ԭ�ӣ���Ӧ���С�6����ԭ�ӡ�8����ԭ�ӡ�4����ԭ�ӣ�����һ��X�ķ�������2����ԭ�ӡ�1��̼ԭ�ӣ�X�Ļ�ѧʽΪCO2��A����������Ԫ�ص���������Ϊ= ��100%��ƫ��������̼����������Ϊ ��100%= 40%����ѧʽ��Ԫ�ط������½ǵ�����Ϊһ��������ԭ�ӵĸ�����N2O...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���ʰ��滯ѧ���꼶�ϲ�5.2�����غ㶨����ϰ�� ���ͣ���ѡ��
ֻ��̼���⡢��Ԫ�ص����ʳ��ȼ�պ�����Ϊ������̼��ˮ����������ͬ�������Ģ�CH4��CH3OH ��C2H4��C2H5OH�ֱ���ȼ�գ���������������ȵ��ǣ� ��
A. �ٺ͢� B. �ٺ͢� C. �ں͢� D. �ۺ͢�
D �������� ֻ��̼���⡢��Ԫ�ص����ʳ��ȼ�պ�����Ϊ������̼��ˮ��������̼��̼��ԭ�Ӹ�����Ϊ1��2��ˮ������ԭ�Ӹ�����Ϊ2��1�����У���CH4��1��̼������2����ԭ�ӣ�4����������2����ԭ�ӣ���������4����ԭ�ӣ���CH3OH��1��̼������2����ԭ�ӣ�4����������2����ԭ�ӣ���ȥ����1��������������3����ԭ�ӣ���C2H4��2��̼������4����ԭ�ӣ�4����������2����ԭ�ӣ���������6����...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���˽̰���꼶��ѧ�ϲ4.2��ˮ�ľ��������ؼ���� ���ͣ������
ij��Ȼ��Ȫˮ��ǩ������������������ˮ����Ҫ�ɷ֣�mg/L����
�����ӣ�0.62��3.25�������ӣ�18.75��29.8�������ӣ�17.8��26.2�������ӣ�l4.2��30.8����������ӣ�41.6��70.2��̼��������ӣ�l84��261�����ԣ�
��1��С����pH��ֽ��øÿ�Ȫˮ��pH����Ϊ________ ������ĸ����
A.7.3 B.13 C.8 D.6
��2��Ҫ�����Ȫˮ��Ӳ������ˮ������________ ��
��3�����������У�����Ȫˮͨ������̿�㣬����̿��������________ ��
C������ˮ������ �������� (1)�ɱ�ǩ��֪�������ӣ�0.62��3.25�������ӣ�18.75��29.8�������ӣ�17.8��26.2�������ӣ�l4.2��30.8����������ӣ�41.6��70.2��̼��������ӣ�l84��261������̼��������ӣ��������ԣ�����pH��ֽ��õ�pHֻ����������ֻ��Cѡ��������⣻(2)����˿�Ȫˮ��Ӳˮ������ˮ���õ������Ƿ���ˮ���������ˮ����ĭ�������ˮ����...�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�������滯ѧ����3.3ȼ�����������ԭ�� ��ϰ ���ͣ���ѡ��
��ѧ�������������ᷢչ�����˾��ס�����֪ʶ���ɶ���ȷ��һ���ǣ� ��
A�����ʶ | B�����ʼ��� |
ϴ�Ӽ�ϴ����---�黯���� ����ȱпԪ��---��״���״� | ʳ������������---Ʒ�� ��ë��������---���� |
C����ȫ��ʶ | D���������� |
�������Ż�---���Ը��Ϲ������ �����̺ܴ�ʱ---��ʪë����ס�ڱ� | ���ٰ�ɫ��Ⱦ---��ʹ��������Ʒ ����������Ⱦ---��ʹ�û�ʯȼ�� |
A. A B. B C. C D. D
C �������� A��ϴ�Ӽ������۾����黯���ã�����ȱ��п���Ỽ��״���״�ѡ�����B��ʳ�κ��������ƶ�����ζ�������������ƶ������к�������Ʒ������ѡ�����C�����Ϲ����ܹ������������Ӷ��ﵽ����Ŀ�ģ���ʪë����ס�ڱǼȿ��Խ��£��ֿ����赲�ҳ�����ѡ����ȷ��D����ʹ�����Ͽ��Լ��ٰ�ɫ��Ⱦ�����Dz�ʹ�û�ʯȼ���ڵ�ǰ������Dz���ʵ�ģ���ѡ�����ѡC���鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com