�⣺��1���������裬�ɹ衢��Ԫ����ɣ���ÿ�������к�2��O��1����ԭ�ӣ��ʻ�ѧʽд��SiO
2��
�ʴ�Ϊ��SiO
2��
��2����ʯ����������ˮ��Ӧ�������������ƣ�����ˮ������Ӧ�Ļ�ѧ����ʽΪCaO+H
2O�TCa��OH��
2��
�ʴ�Ϊ��CaO+H
2O�TCa��OH��
2��
��3��1kg��ˮ�Ȼ�������1.2kgˮ��õ��Ȼ�����Һ������Һ�����ʵ���������=

��100%=45.5%��
�ʴ�Ϊ��45.5%��
��4����ΪŨ�����к�ǿ�ĸ�ʴ�ԣ��ճ������к�����Ũ������Ϊ�������
�ʴ�Ϊ��Ũ�����к�ǿ��ʴ�ԣ�
��5�����ڳ�ʪ�Ŀ�������ˮ����������һϵ�и��ӵĻ�ѧ�仯�����⣬���ƶ���Ϊ��˫�������Ļ�ԭ����Ӧ���տ����е�ˮ���������ʳơ�˫��������˫�������Ƿ���Чȡ�����Ƿ��Ժ������ۣ���ˣ�ȡʹ����һ��ʱ��ġ�˫��������Ʒ���μ�ϡ���ᣬ�������ݲ�����˵������������Ч���������ݣ�˵����������ȫ��Ӧ������Ч��
�ʴ�Ϊ��������ˮ��ȡ������ϡ���ᣬ�������ݲ���������Ч����֮����Ч��
��������1�������ʵ����ƶ���������ж������ɹ衢��Ԫ����ɣ���ÿ�������к�2��O��1����ԭ�ӣ��ݴ�д�����ʶ�������Ļ�ѧʽ��
��2����ʯ��Ϊ�����Ƶ��׳ƣ�����ˮ�������Ϸ�Ӧ�����������ƣ�
��3���������ʵ���������=
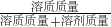
��100%������1kg��ˮ�Ȼ�������1.2kgˮ��õ��Ȼ�����Һ�����ʵ�����������
��4������Ũ������к�ǿ�ĸ�ʴ�ԣ������ճ�������ȴ������������ʪ����
��5�����������������������ˮ��������ͬ���õĽ�����ƶϡ�˫���������ʣ����顰˫�������Ƿ�����Ч�������Ƿ������ۣ��ɲ�ȡ�μ�ϡ����۲����ݵķ���֤�����۵Ĵ��ڣ�
������һ�����֪ʶ���������ܵĿ��飬�漰��ѧʽ����ѧ����ʽ����д���������������ļ��㼰ʵ���Լ�𣬽��ʱӦ����������Ҫ��Ϊһ̸��

 ��100%=45.5%��
��100%=45.5%��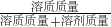 ��100%������1kg��ˮ�Ȼ�������1.2kgˮ��õ��Ȼ�����Һ�����ʵ�����������
��100%������1kg��ˮ�Ȼ�������1.2kgˮ��õ��Ȼ�����Һ�����ʵ�����������

 ÿ��10���ӿ�����������������ϵ�д�
ÿ��10���ӿ�����������������ϵ�д�