

|
 2H2O2 2H2O + O2��
2H2O2 2H2O + O2�� 2H2O + O2����������˫��ˮ������¶˵ĵ����ܿڻᱻҺ�⣬��������ܶȶ���С���������Ĺܿ��ݳ�������ر�K1,��K2��
2H2O + O2����������˫��ˮ������¶˵ĵ����ܿڻᱻҺ�⣬��������ܶȶ���С���������Ĺܿ��ݳ�������ر�K1,��K2��

 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| | ʵ�鷽�� | ���� |
| A | ��ʢװCO��������ƿ�м���NaOH��Һ���۲쵽ƿ����� | ˵��CO2����NaOH��Ӧ |
| B | ľ̿�ڿ����������о���ȼ�� | ˵��������֧��ȼ�� |
| C | ij��ɫ��ĩ�����ֽⷴӦ��ֻ���ɵ��ʹ������� | �÷�ĩ����Ԫ�غ�Ԫ����� |
| D | ����ɫʯ����Һ��ͨ��CO2��ʯ����Һ��� | CO2������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

| ʵ���� | �� | �� | �� |
| ��Ӧ�� | 5%H2O2 50 mL | 5%H2O2 | 3%H2O250 mL |
| ������� | 0.5gCuO | 0.5gMnO2 | 0.5gMnO2 |
| �ռ������O2����ʱ�� | 105 s | 45s | 78 s |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ʵ�鲽�� | Ԥ����������� |
| ��1�� | |
| ��2�� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
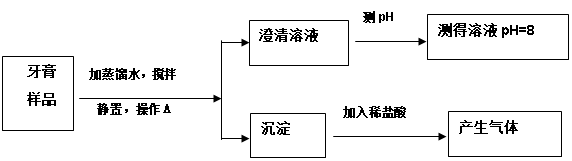
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

| ̽��Ŀ�� | ̽������ | Ԥ������ |
| ������Һ�е�CO32- | �٣�ȡ������Һ���Թ��У��μ�������CaCl2�Լ� | |
| ֤����Һ���д�NaOH | �ڣ���ʵ��ٹ��˺�������Һ�еμӷ�̪��Һ | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

| �¶�(��) | 10 | 20 | 40 | 60 | 80 | 100 |
| �ܽ��(g/100gˮ) | 64 | 85 | 138 | 203 | 285 | 376 |


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
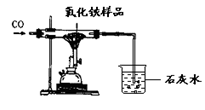
| | ��Ӧǰ | ��������ȫ��Ӧ�� |
| A�� | �����ܺ���������Ʒ������43.7g | �����ܺ��������41.3g |
| B�� | �ձ��ͳ���ʯ��ˮ������180g | �ձ����ձ������ʵ�����186.8g |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com