
ĻÖÓŠŅ»¶ØÖŹĮæŗ¬ÓŠÉŁĮæÄąÉ³µČ²»ČÜŠŌŌÓÖŹŗĶÉŁĮæNa2SO4£¬MgCl2£¬CaCl2µČæÉČÜŠŌŌÓÖŹµÄ“ÖŃĪѳʷ£¬Ä³ŹµŃ銔×éĄūÓĆ»ÆѧŹµŃéŹŅ³£ÓĆŅĒĘ÷¶Ō“ÖŃĪѳʷ½ųŠŠĢį“棬Ģį“æ²½ÖčČēĻĀ£ŗ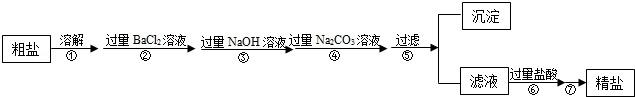
Ēėøł¾ŻĢį“æ²½Öč»Ų“šĻĀĮŠĪŹĢā£®
£Ø1£©²½Öč¢ßµÄ²Ł×÷Ćū³ĘĪŖ £®
£Ø2£©ĒėŠ“³öŹµŃé²½Öč¢ŚÖŠĖłÉę¼°µÄ»Æѧ·½³ĢŹ½ £®
£Ø3£©²½Öč¢ŽÖŠ¼ÓČė¹żĮæŃĪĖįµÄÄæµÄŹĒ £®
£Ø4£©²½Öč¢ŚŗĶ²½Öč¢Ü £ØĢī”°æÉŅŌ”±»ņ”°²»æÉŅŌ”±£©µßµ¹£¬ĄķÓÉŹĒ £®
£Ø5£©¼ģŃé²½Öč¢ÜÖŠNa2CO3ČÜŅŗŅŃ¹żĮæµÄ·½·ØŹĒ £®
£Ø6£©¼ÓµāŹ³ŃĪĻą¹ŲŠÅĻ¢ČēĶ¼ĖłŹ¾£®
Ź³ŃĪÖŠµÄµāĖį¼Ų£ØKIO3£©ŌŚĖįŠŌĢõ¼žĻĀ£¬æÉŅŌ½«µā»Æ¼Ų£ØKI£©±ä³Éµā£ØI2£©£¬»Æѧ·½³ĢŹ½ČēĻĀ£ŗ
KIO3+5KI+6HCl=6KCI+3I2+3H2O
¢ŁĻņ×°ÓŠµā»Æ¼ŲŗĶµķ·Ū»ģŗĻŅŗµÄŹŌ¹ÜÖŠ£¬µĪČėĻ”ŃĪĖį½«ČÜŅŗĖį»Æ£¬ŌŁ¼ÓČėŹ³ŃĪ£¬ČōŹ³ŃĪÖŠÓŠµā»Æ¼Ų£¬Ōņ¼ÓČėŹ³ŃĪŗóµÄŹµŃéĻÖĻó £®
¢ŚŠ”ĒæĶ¬Ń§Óū²ā¶Ø¼ÓµāŃĪÖŠµāŌŖĖŲµÄÖŹĮæ·ÖŹż£¬ŹµŃé²½ÖčČēĻĀ£ŗČ”10gŹ³ŃĪѳʷӌŹŌ¹ÜÖŠ¼ÓĖ®Čܽā£¬¼ÓČė¹żĮæKIµÄŗĶµķ·Ū»ģŗĻČÜŅŗ£¬ŌŁµĪČėĻ”ŃĪĖį½«ČÜŅŗĖį»ÆŹ¹Ęä³ä·Ö·“Ó¦ŗ󣬵÷½ŚČÜŅŗ³ŹÖŠŠŌ£¬ŌŁĻņŹŌ¹ÜÖŠµĪ¼ÓĮņ“śĮņĖįÄĘČÜŅŗ£ØNa2S2O3£©£¬·¢Éś»Æѧ·“Ó¦·½³ĢŹ½ĪŖ£ŗ2Na2S2O3+I2ØTNa2S4O6+2NaI
µ±¼ÓČėÖŹĮæ·ÖŹżĪŖ0.237%Na2S2O3ČÜŅŗ2gŹ±£¬I2Ē”ŗĆ·“Ó¦ĶźČ«£¬Ķعż¼ĘĖćÅŠ¶ĻøĆŹ³ŃĪѳʷŹĒ·ńŗĻøń£ØŅŃÖŖNa2S2O3µÄĻą¶Ō·Ö×ÓÖŹĮæĪŖ158£®ĒėŠ“³ö¼ĘĖć¹ż³Ģ£©£®
£Ø1£©Õō·¢½į¾§
£Ø2£©BaCl2+Na2SO4ØTBaSO4”ż+2NaCl£®
£Ø3£©³żČ„¹żĮæµÄĒāŃõ»ÆÄĘŗĶĢ¼ĖįÄĘ£®
£Ø4£©²»æÉŅŌ£»Čē¹ūµßµ¹£¬ĪŽ·Ø³żČ„¹żĮæµÄĀČ»Æ±µ£®
£Ø5£©Č”ÉĻ²ćĒåŅŗÓŚŹŌ¹ÜÖŠ£¬µĪ¼ÓĀČ»ÆøĘČÜŅŗ£¬Čē¹ū³öĻÖ°×É«³Įµķ£¬ĖµĆ÷Ģ¼ĖįÄĘČÜŅŗŅŃ¾¹żĮæ
£Ø6£©²»ŗĻøń
½āĪöŹŌĢā·ÖĪö£ŗ£Ø1£©²½Öč¢ßµÄ²Ł×÷Ćū³ĘĪŖÕō·¢½į¾§£®
¹ŹĢī£ŗÕō·¢½į¾§£®
£Ø2£©ŹµŃé²½Öč¢ŚÖŠÉę¼°ĀČ»Æ±µŗĶĮņĖįÄĘ·“Ó¦£¬ĀČ»Æ±µŗĶĮņĖįÄĘ·“Ó¦ÄÜÉś³ÉĮņĖį±µŗĶĀČ»ÆÄĘ£¬»Æѧ·½³ĢŹ½ĪŖ£ŗBaCl2+Na2SO4ØTBaSO4”ż+2NaCl£®
¹ŹĢī£ŗBaCl2+Na2SO4ØTBaSO4”ż+2NaCl£®
£Ø3£©²½Öč¢ŽÖŠ¼ÓČė¹żĮæŃĪĖįµÄÄæµÄŹĒ³żČ„¹żĮæµÄĒāŃõ»ÆÄĘŗĶĢ¼ĖįÄĘ£®
¹ŹĢī£ŗ³żČ„¹żĮæµÄĒāŃõ»ÆÄĘŗĶĢ¼ĖįÄĘ£®
£Ø4£©Čē¹ūµßµ¹£¬ĪŽ·Ø³żČ„¹żĮæµÄĀČ»Æ±µ£®
¹ŹĢī£ŗ²»æÉŅŌ£»Čē¹ūµßµ¹£¬ĪŽ·Ø³żČ„¹żĮæµÄĀČ»Æ±µ£®
£Ø5£©¼ģŃé²½Öč¢ÜÖŠNa2CO3ČÜŅŗŅŃ¹żĮæµÄ·½·ØŹĒ£ŗČ”ÉĻ²ćĒåŅŗÓŚŹŌ¹ÜÖŠ£¬µĪ¼ÓĀČ»ÆøĘČÜŅŗ£¬Čē¹ū³öĻÖ°×É«³Įµķ£¬ĖµĆ÷Ģ¼ĖįÄĘČÜŅŗŅŃ¾¹żĮ森
¹ŹĢī£ŗČ”ÉĻ²ćĒåŅŗÓŚŹŌ¹ÜÖŠ£¬µĪ¼ÓĀČ»ÆøĘČÜŅŗ£¬Čē¹ū³öĻÖ°×É«³Įµķ£¬ĖµĆ÷Ģ¼ĖįÄĘČÜŅŗŅŃ¾¹żĮ森
£Ø6£©ÓÉKIO3+5KI+6HCl=6KCI+3I2+3H2OŗĶ2Na2S2O3+I2ØTNa2S4O6+2NaIæÉÖŖ£¬IŗĶNa2S2O3µÄ¶ŌÓ¦¹ŲĻµĪŖ£ŗI”ś6Na2S2O3£¬
Éč10gŹ³ŃĪѳʷ֊µāŌŖĖŲµÄÖŹĮæĪŖX£¬
2g0.237%µÄNa2S2O3ČÜŅŗÖŠNa2S2O3µÄÖŹĮæĪŖ£ŗ2g”Į0.237%=0.00474g£¬
I”ś6Na2S2O3£¬
127 948
X 0.00474g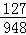 =
=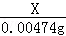
X=0.000635g£¬
10gŹ³ŃĪѳʷ֊µāŌŖĖŲµÄÖŹĮæŹĒ0.000635g£¬1000gŹ³ŃĪѳʷ֊µāŌŖĖŲµÄÖŹĮæĪŖ£ŗ0.000635g”Į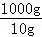 =0.0635g=63.5mg£¬
=0.0635g=63.5mg£¬
ÓėĶ¼ÖŠµÄŠÅĻ¢±Č½Ļ£¬øĆŹ³ŃĪѳʷ²»ŗĻøń£®
æ¼µć£ŗĀČ»ÆÄĘÓė“ÖŃĪĢį“棻¹żĀĖµÄŌĄķ”¢·½·Ø¼°ĘäÓ¦ÓĆ£»ŃĪµÄ»ÆѧŠŌÖŹ£»øł¾Ż»Æѧ·“Ó¦·½³ĢŹ½µÄ¼ĘĖć£»¼ų±šµķ·Ū”¢ĘĻĢŃĢĒµÄ·½·ØÓėµ°°×ÖŹµÄŠŌÖŹ£®
µćĘĄ£ŗ±¾ĢāÖ÷ŅŖæ¼²é“ÖŃĪĢį“æµÄ¹ż³ĢŗĶøł¾Ż»Æѧ·½³ĢŹ½½ųŠŠ¼ĘĖć£¬¼ĘĖ揱Ņ»¶ØŅŖČĻÕę׊Ļø£¬±ÜĆā³ö“ķ£®


 æŚĖćŠÄĖćĖŁĖćÓ¦ÓĆĢāĻµĮŠ“š°ø
æŚĖćŠÄĖćĖŁĖćÓ¦ÓĆĢāĻµĮŠ“š°ø Ķ¬²½ĶŲÕ¹ŌĶĮĻµĮŠ“š°ø
Ķ¬²½ĶŲÕ¹ŌĶĮĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¼ĘĖćĢā
ijĮņĖį³§·ĻĖ®ÖŠŗ¬ÓŠÉŁĮæĮņĖį£¬ĪŖ“ļ±źÅÅ·Å£¬¼¼ŹõŌ±Š”ÕŶŌ·ĻĖ®ÖŠĮņĖįµÄŗ¬Įæ½ųŠŠ¼ģ²ā£®
£Ø1£©ÅäÖĘČÜŅŗ£ŗ ÓūÅäÖĘČÜÖŹÖŹĮæ·ÖŹżĪŖ4%µÄNaOHČÜŅŗ100g£¬ŠčŅŖNaOH¹ĢĢå g£¬Ė® ml£ØĖ®µÄĆܶČĪŖ1g/cm3£©£»
£Ø2£©¼ģ²ā·ÖĪö£ŗ Č”·ĻĖ®ŃłĘ·98g£¬ĻņĘäÖŠÖšµĪ¼ÓČėNaOHČÜŅŗÖĮĒ”ŗĆĶźČ«·“Ó¦Ź±£¬ĻūŗÄ4%µÄNaOHČÜŅŗ20g”££Ø¼ŁÉč·ĻĖ®ÖŠĘäĖü³É·Ö¾ł²»ŗĶNaOH·“Ó¦£»·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ£ŗ2NaOH+H2SO4ØTNa2SO4+2H2O£©£» ŹŌ¼ĘĖć·ĻĖ®ÖŠĮņĖįµÄÖŹĮæ·ÖŹż£ØŠ“³ö¼ĘĖć¹ż³Ģ£©£®
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¼ĘĖćĢā
ĪŅŹŠĀ®½ĻŲŠ”°ü×ÆČ„Äź·¢ĻÖŅ»“óŠĶĢśæó£¬Ö÷ĢåĪŖ“ÅĢśæó£®Ņ±Į¶“ÅĢśæóµÄ·“Ó¦£ŗ4CO+Fe3O4 3Fe+4CO2£®ÓĆ100tŗ¬Fe3O480%µÄ“ÅĢśæóŹÆ£¬ĄķĀŪÉĻŅ±Į¶ŗ¬ŌÓÖŹ4%µÄÉśĢś¶ąÉŁ£æ£Ø“š°ø±£ĮōŅ»Ī»Š”Źż£©
3Fe+4CO2£®ÓĆ100tŗ¬Fe3O480%µÄ“ÅĢśæóŹÆ£¬ĄķĀŪÉĻŅ±Į¶ŗ¬ŌÓÖŹ4%µÄÉśĢś¶ąÉŁ£æ£Ø“š°ø±£ĮōŅ»Ī»Š”Źż£©
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¼ĘĖćĢā
½«50gĀČ»ÆøĘČÜŅŗÓė77gĢ¼ĖįÄĘČÜŅŗ»ģŗĻŗó£¬Ē”ŗĆĶźČ«·“Ó¦£¬¹żĀĖ”¢Ļ“µÓ”¢ŗęøÉŗó£¬µĆµ½10g°×É«¹ĢĢ壮Ēė¼ĘĖć£ØŠ“³ö¼ĘĖć¹ż³Ģ£©£ŗ
£Ø1£©øĆ50gĀČ»ÆøĘČÜŅŗÖŠČÜÖŹµÄÖŹĮ棻
£Ø2£©¹żĀĖŗóĖłµĆČÜŅŗÖŠČÜÖŹµÄÖŹĮæ·ÖŹż£®
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¼ĘĖćĢā
ijŠ£»ÆѧŠĖȤŠ”×éĪŖ²ā¶ØæÕĘųÖŠ¶žŃõ»ÆĮņµÄŗ¬Į棬ÓĆNaOHČÜŅŗĪüŹÕSO2£¬·“Ó¦µÄ·½³ĢŹ½ČēĻĀ£ŗ2NaOH+SO2ØTNa2SO4+H2O£®ÓĆNaOHČÜŅŗĪüŹÕ100LŅŃ³żČ„CO2µÄæÕĘųѳʷ£¬ČÜŅŗÖŹĮæŌö¼ÓĮĖ0.64g£®ŅŃÖŖ“ĖŹ±æÕĘųµÄĆܶČĪŖ1.3g/L£¬Ēó£ŗ
£Ø1£©±»ĪüŹÕµÄSO2µÄÖŹĮ森
£Ø2£©·¢Éś·“Ó¦µÄNaOHµÄÖŹĮ森
£Ø3£©æÕĘųÖŠSO2µÄÖŹĮæ·ÖŹż£Ø¼ĘĖć½į¹ū¾«Č·µ½0.01%£©£®
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¼ĘĖćĢā
ĖÉÓĶ“¼£ØC10H18O£©ŹĒŅ»ÖÖµ÷ĻćĻć¾«£¬¶ą“ęŌŚÓŚĖɽŚÓĶ”¢·¼ÕĮÓĶ”¢ÓńŹ÷ÓĶ”¢³Č»ØÓĶÖŠ£®Ēė»Ų“š£ŗ
£Ø1£©ĖÉÓĶ“¼ÖŠĒāŌŖĖŲŗĶŃõŌŖĖŲµÄÖŹĮæ±ČŹĒ_________ £»
£Ø2£©ĖÉÓĶ“¼µÄĻą¶Ō·Ö×ÓÖŹĮæŹĒ_________ £»
£Ø3£©15.4gĖÉÓĶ“¼Ėłŗ¬Ģ¼ŌŖĖŲÖŹĮæÓė_________ gĘĻĢŃĢĒ£ØC6H12O6£©Ėłŗ¬Ģ¼ŌŖĖŲÖŹĮæĻąµČ£®
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¼ĘĖćĢā
ŅŃÖŖ½šŹōÄĘÄÜÓėĖ®·¢ÉśČēĻĀ·“Ó¦£ŗ2Na+2H2OØT2NaOH+H2”ü£¬Čō°Ń4.6g½šŹōÄĘĶ¶Čėµ½Ź¢ÓŠ×ćĮæĖ®µÄÉÕ±ÖŠ£ØČēĶ¼£©£¬³ä·Ö·“Ó¦ŗóÉÕ±ÖŠŹ£ÓąČÜŅŗÖŹĮæŹĒ40g£¬Ēė¼ĘĖć£ŗ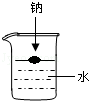
£Ø1£©Éś³ÉNaOHµÄÖŹĮ森
£Ø2£©·“Ó¦ŗóĖłµĆNaOHČÜŅŗµÄČÜÖŹÖŹĮæ·ÖŹż£®
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¼ĘĖćĢā
Ēā»ÆøĘ£ØCaH2£©¹ĢĢåŹĒµĒɽ¶ÓŌ±³£ÓƵÄÄÜŌ“Ģį¹©¼Į£®Ä³»ÆѧŠĖȤŠ”×éÄāÓĆČēĶ¼1ĖłŹ¾µÄ×°ÖĆÖʱøĒā»ÆøĘ£¬Ęä·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ£ŗCa+H2 CaH2£®
CaH2£®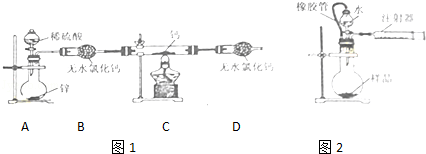
£Ø1£©CaH2ÖŠøĘŗĶĒāŌŖĖŲµÄ»ÆŗĻ¼Ū·Ö±šĪŖ”” ””£¬×°ÖĆÖŠµÄĪŽĖ®ĀČ»ÆøĘøÉŌļ×°ÖĆŅ²æÉÓĆ”” ””Ą““śĢę£®
£Ø2£©ĄūÓĆøĆ×°ÖĆ½ųŠŠŹµŃ飬²½ÖčČēĻĀ£ŗ¼ģ²é×°ÖƵÄĘųĆÜŠŌŗó×°ČėŅ©Ę·£¬“ņæŖ·ÖŅŗĀ©¶·»īČū£ŗ”” ””£ØĒė°“ÕżČ·µÄĖ³ŠņĢīČėĻĀĮŠ²½ÖčµÄŠņŗÅ£©£®
¢Ł¼ÓČČ·“Ó¦Ņ»¶ĪŹ±¼ä ¢ŚŹÕ¼ÆĘųĢå²¢¼ģŃéĘä“æ¶Č ¢Ū¹Ų±Õ·ÖŅŗĀ©¶·»īČū ¢ÜĶ£Ö¹¼ÓČČ£¬³ä·ÖĄäČ“
£Ø3£©ĪŖĮĖČ·ČĻ½ųČė×°ÖĆCµÄĒāĘųŅŃ¾øÉŌļ£¬Ó¦ŌŚB”¢CÖ®¼äŌŁĮ¬½ÓŅ»×°ÖĆX£¬×°ÖĆXÖŠ¼ÓČėµÄŹŌ¼ĮŹĒ”” ””£®ČōĒāĘųĪ“³ä·ÖøÉŌļ£¬×°ÖĆXÖŠµÄĻÖĻóĪŖ”” ””£®
£Ø4£©ĪŖĮĖ²āĮæÉĻŹöŹµŃéÖŠÖʵƵÄĒā»Æøʵēæ¶Č£¬øĆŠ”×é³ĘČ”mgĖłÖʵĆѳʷ£¬°“ČēĶ¼2ĖłŹ¾×°ÖĆ½ųŠŠ²ā¶Ø£®ŠżæŖ·ÖŅŗĀ©¶·»īČū£¬·“Ó¦½įŹųŗó³ä·ÖĄäČ“£¬×¢ÉäĘ÷»īČūÓÉ·“Ó¦Ē°µÄV1mLæĢ¶Č“¦±ä»Æµ½V2mLæĢ¶Č“¦£ØV2£¼V1£¬ĘųĢåĆܶČĪŖdg/mL£©
¢ŁĻš½ŗ¹ÜµÄ×÷ÓĆĪŖ£ŗa£®”” ””£»b£®”” ””£®
¢ŚŠżæŖ·ÖŅŗĀ©¶·»īČūŗ󣬳ż·¢ÉśCaH2+H2OØTCa£ØOH£©2+H2”üµÄ·“Ó¦Ķā£¬»¹×īÓŠæÉÄÜ·¢ÉśµÄ·“Ó¦ĪŖ”” ””£®
¢ŪÓĆw±ķŹ¾Ēā»Æøʵēæ¶Č£¬ĒėÓĆŅ»øöµČŹ½±ķŹ¾³öd”¢V1”¢V2ŗĶwÖ®¼äµÄ¹ŲĻµ”””” £®
¢ÜøĆŠ”×éŅŅĶ¬Ń§ČĻĪŖČ„µōĮ¬½ÓµÄ×¢ÉäĘ÷£¬Ņ²ŅŖ¼ĘĖć³öĒā»Æøʵēæ¶Č£®ĖūĶ¬Ńł³ĘČ”mgѳʷ£¬¼ÓČėÉÕĘæÖŠŗó³ĘČ”·“Ó¦Ē°µÄÖŹĮæĪŖm1g£¬·“Ó¦ŗóµÄÖŹĮæĪŖm2g£®ŅŅĶ¬Ń§±ķŹ¾³öµÄm£¬m1£¬m2ŗĶwÖ®¼äµÄ¹ŲĻµµÄµČŹ½ĪŖ”” £®
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¼ĘĖćĢā
½«20gŗ¬ŌÓÖŹ£ØŌÓÖŹ²»ČÜÓŚĖ®£¬Ņ²²»ČÜÓŚĖį£©µÄŠæѳʷ£¬ÓėŅ»¶ØÖŹĮæµÄĻ”ĮņĖįĒ”ŗĆĶźČ«·“Ó¦£¬ĖłµĆČÜŅŗÖŹĮæ80.5g£¬Ķ¬Ź±Éś³ÉĒāĘų0.4g£¬ŹŌ¼ĘĖć£ŗ
£Ø1£©ŃłĘ·ÖŠŠæµÄÖŹĮæĪŖ£ŗ g”£
£Ø2£©ĶźČ«·“Ó¦ŗóĖłµĆČÜŅŗÖŠČÜÖŹµÄÖŹĮæ·ÖŹż£ØŠ“³ö¼ĘĖć¹ż³Ģ£©”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com